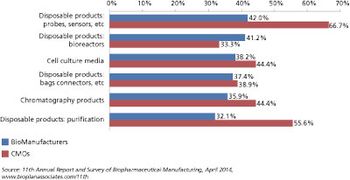
Biomanufacturers are seeking more innovation and expertise from their CMO partners.

Biomanufacturers are seeking more innovation and expertise from their CMO partners.
Whether outsourcing or developing cell therapies in-house, success demands a focus on quality, cost of goods, and sustainability from the start.

CMO executives share their opinions on where outsourcing is going and what is driving market change.

As CMOs shed their old toll processing role, sponsors can expect the right questions, proactive communication, and a firm grasp of risk management and tech transfer from contract service providers.
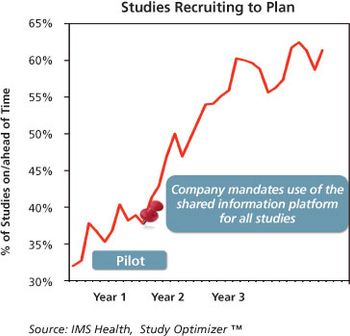
Data sharing and a common data model can improve CRO partnerships and trial results.
After launching a new mammalian cell platform, FUJIFILM Diosynth Biotechnologies U.S.A., has acquired fast-track vaccine manufacturing knowhow and a major presence in Texas’ emerging biocorridor with Kalon, its first acquisition.

Better equipment and automated processes will be key. So, too, will the right way of approaching outsourcing relationships.