
ePT--the Electronic Newsletter of Pharmaceutical Technology
Rockville, MD (Dec. 12)-The US Food and Drug Administration is proposing amendments to its final rule regarding labeling requirements for convenience-size over-the-counter human drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Rockville, MD (Dec. 12)-The US Food and Drug Administration is proposing amendments to its final rule regarding labeling requirements for convenience-size over-the-counter human drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Dublin, OH (Nov. 14)-A half-year Cardinal Health study of radio-frequency identification (RFID) tags "under real-world conditions has demonstrated that the technology has real promise to provide an added layer of safety," according Renard Jackson, the company's vice-president and general manager of global packaging services, in a prepared statement.
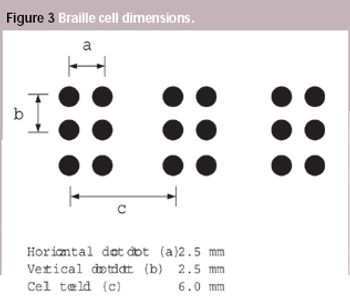
Pharmaceutical Technology Europe
A European Commission directive requires suppliers to incorporate information on medicines in formats suitable for blind and partially sighted people, although the associated standard has not been introduced yet. This article looks at some of the issues that will need to be addressed when the standard is adopted.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Robin Hwang, a senior principal scientist at Amgen (Thousand Oaks, CA), led the team that developed the first commercial disposable auto-injector for a biopharmaceutical: a prefilled three-step "SureClick" for delivering Enbrel (etanercept), a treatment for autoimmune diseases.

Nondestructive testing takes many forms and is gaining favor because it saves time and prevents costly product loss.

Pharmaceutical Technology Europe
It will remain to be seen whether pharmaceutical companies will commit lock, stock and barrel to being green.

A hybrid system using paper and electronic pedigrees will be needed.

Pharmaceutical Technology Europe
RFID is viewed by many, including FDA, as a technology with strong potential for carrying the mass serialization data needed to track and trace product and to create pedigree records.

Disappointed in progress thus far, the US Food and Drug Administration wants pharmaceutical manufacturers to make a greater effort to combat counterfeit products and recommends that they "move quickly" to implement radio-frequency identification technology.

How does the latest agency task force report resonate for pharma and radio-frequency identification?

ePT--the Electronic Newsletter of Pharmaceutical Technology
A group of researchers from Georgia Institute of Technology (Atlanta, GA) are using high-throughput ionization techniques to identify and measure the ingredients in counterfeit drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration?s Counterfeit Drug Task Force (Rockville, MD, www.fda.gov) is recommending regulatory actions and the implementation of new technologies for reducing the risk of counterfeit drugs entering the United States. The group has followed up on its original 2004 report, in which it outlined the framework for protecting the public from counterfeit medicines, and an updated 2005 report with a third document encouraging electronic pedigrees, improved traceability in the drug supply chain, and the adoption of radio-frequency identification (RFID) tools.

I always suspected that our purchasing manager had agreed to this just to save money . . .

Interphex provided an opportunity to examine the latest pharmaceutical packaging concepts and packaging machines.

Bringing Exubera to market requires extensive collaboration by Pfizer, Nektar Therapeutics, West Pharmaceutical Services Tech Group, and Bespak.

Pharmaceutical Technology Europe
This article looks at the different types of marking and coding techniques used in the pharmaceutical supply chain and their role in helping to prevent counterfeiting.

The need to curb drug counterfeiting is spurring development of track-and-trace and product authentication technologies.

Despite worries that industry is slow to adopt anticounterfeiting technologies, the 2006 Interphex program is rife with new methods for securing the supply chain.

Pharmaceutical Technology Europe
The right choice [of coding and marking technology] depends upon the company's top priorities regarding legibility, cost, speed, ease of use, cleanliness and security.

GSK Begins RFID Pilot Program

ePT--the Electronic Newsletter of Pharmaceutical Technology
RFID and the Future of Pharmaceutical Supply Chains

PDA's Technical Report No. 39 provides guidance for protecting temperature-sensitive products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Pharmaceutical Industry has been slow in adopting radio frequency technology (RFID) to help control diversion and counterfeiting, according to a recent study by ABI Research (Oyster Bay, NY, www.abiresearch.com). In fact, only 10 drug products are expected to be shipped with RFID tags or smart chips embedded in the labeling in the coming year.
Pharmaceutical Technology Europe
The type of robot used for placing and stacking the BFS cards is important. Conventional multi-axes designs have limited flexibility, often combined with high inertia that limits operating speeds.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer Combats Counterfeiters with RFID