
QbD paradigm advances process understanding in development and manufacturing.

QbD paradigm advances process understanding in development and manufacturing.

Vaccine development is benefiting from manufacturing advances and support for global health.

Brazil's major vaccine producer innovates with stem-cell research.

Factors for assessing excipient variability, the associated challenges developers need to address to design and manufacture solid oral drug products, and solutions for such challenges are examined.
The use of various documents and guides by the International Pharmaceutical Excipients Council can facilitate the flow of information among excipient manufacturers, distributors, and users.

An alternative chapter has been added to the European Pharmacopoeia for dosage uniformity.

The European Medicines Agency?s Committee for Orphan Medicinal Products (COMP) is seeking to expand its international cooperation in 2013.

FDA has released a list of more than 50 guidance documents planned for 2013.

The latest news from the pharmaceutical regulatory community.

Siegfried Schmitt, a principal consultant with PAREXEL, discusses how the EU's Falsified Medicines Directive will affect US API production.

Quality assurance of biological products is central to India's good distribution practices guidelines.

Prosecutors and regulators challenge manufacturing quality failings likely to cause patient harm.
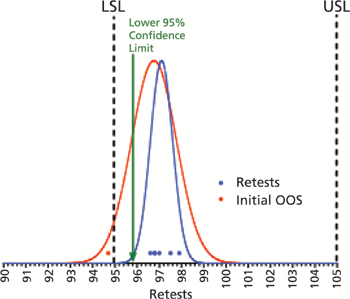
Two methods to evaluate retest data following out-of-specification results.

A thorough investigation of all possible causes of deviations should be performed.

The US Pharmacopeial Convention contiues to make modernization of standards a top priority in 2013.

Protecting patients from counterfeit medicines is a pressing issue facing governments and the pharmaceutical industry.

Recent activity in standards-setting organizations has raised interest in the impact of testing for impurities that may enter the product before it is mined or harvested or even due to intentional use of some reagents.

Adherence to GMP in API manufacturing is crucial in determining the safety of drug products.

Two general chapters on elemental impurities? limits and procedures are to become official Feb. 1, 2013, with implementation proposed for May 1, 2014.

EMA announces collaboration with Israel, and USP updates chapter on elemental impurities.

Ben Venue Laboratories announced that it has voluntarily entered into a consent decree with FDA over violations of cGMP.

FDA has issued a Warning Letter to Hameln Pharmaceuticals GmbH after observing "significant" cGMP violations for finished pharmaceuticals at the company?s facility in Hameln, Germany.

FDA has released Guidance for Industry: Abuse-Deterrent Opioids?Evaluation and Labeling, Draft Guidance

Eli Lilly expects overall revenue growth in the coming year.

FDA celebrates 30 years of the Orphan Drug Act.