
Manufacturing and in-depth characterization provide basis for demonstrating product equivalence.

Manufacturing and in-depth characterization provide basis for demonstrating product equivalence.

Even when all is well at the facility, one must expect the worst while braving the elements.
The European Medicines Agency has added granularity to its biosimilars approval pathway by releasing a guideline on biosimilar monoclonal antibodies (mAbs).

The government of Turkey is drawing up a program in coordination with the pharmaceutical industry to create ways to make the country a regional production center for pharmaceuticals serving Europe, Central Asia, and the Middle East.

The US Pharmacopeia's revised General Chapters on elemental impurity limits and testing procedures are set to take effect in December 2012.

The Pharmaceutical Inspection Co-operation Scheme (PIC/S) has finalized a risk-based inspection planning tool for inspectorates to use in applying science- and risk-based principles to planning GMP inspections.
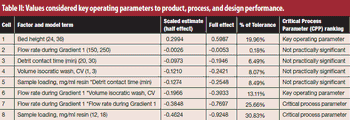
Critical process parameters (CPPs) and their associated process controls are crucial to drug development and process validation and to the evaluation of every manufacturing unit operation.
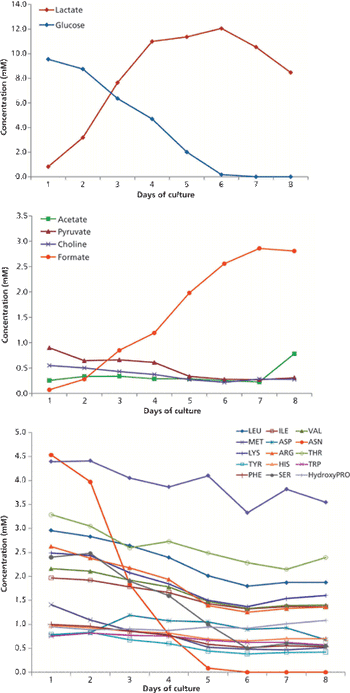
The author discusses current expectations in bioprocessing and lays a framework for using NMR to enhance a QbD approach.

New England Compounding Center gets 483 after linked fungal meningitis outbreak.

FDA announces Coalition for Accelerating Standards and Therapies and Commissioner Hamburg comments on meningitis outbreak.

EMA releases guideline on medicinal products for the treatment of schizophrenia.

New US Pharmacopeial Convention standards provide a universal approach to organizing labels for prescription containers dispensed by US pharmacists in an effort to improve patient understanding.

Managing risk in biopharmaceutical operations is of utmost importance for patient protection.

New European Pharmacopoeia chapter aims to resolve problems with applying the harmonized UDU test to large sample sizes.
IQ Consortium representatives explore and define common industry approaches and practices for applying GMPs in early development.

Will the next US President support the backbone of our industry?
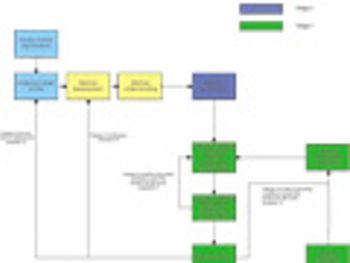
The authors describe how traditional approaches to analytical method and validation may benefit from alignment with quality-by-design concepts.

This article provides a comparison of Rx-360, EXCiPACT and IPEA, available to pharmaceutical manufacturers for the purpose of auditing excipient suppliers and ensuring drug efficacy and patient safety.

Foreign firms struggle against stricter patent laws, but all is not lost.

Real-time experimentation may offer continuous process improvement.

Only the strong survive when it comes to pharmaceutical packaging and shipping.
This study examines the effect and interaction of variations in hypromellose physicochemical properties.

Working together affords many unseen opportunities for pharmaceutical innovation.

The promise of the Generic Drug User Fee Amendments of 2012 is to end multiyear reviews of new generic drugs and the ever-growing queue of pending applications.

FDA launches BeSafeRx program and releases guidance for industry.