
FDA funds research to further development of innovative generics, while working to address review and approval issues.

FDA funds research to further development of innovative generics, while working to address review and approval issues.

Recent draft guidance from FDA on contract manufacturing and quality agreements highlights the importance of such agreements and define the roles and responsibilities of each party to be in keeping with quality risk-management principles.

Rx-360, a pharmaceutical industry supply-chain consortium, is advancing approaches between pharmaceutical companies, suppliers, and contract manufacturers to better secure the pharmaceutical supply chain.
Increased pharmaceutical trade creates new challenges for regulatory oversight.

Depomed, Inc. announced that FDA has accepted for filing a new drug application (NDA) from Mallinckrodt for MNK-795.

The precautionary recall follows manufacturing deficiencies identified at the site in India.

Europe prepares for inclusion of Croatia in EMA activities.

Programs assist in the fast track of drugs for serious conditions.

Brazil offers opportunities and challenges for global pharmaceutical companies.

Falsified Medicines Directive requires imported APIs to have written confirmation of GMP standards.

David Elder, vice-president, technical at PAREXEL, discusses the best way to productively participate in a regulatory meeting with FDA.

European governments are under pressure to take regulatory action, but solving the problem of medicine shortages is not as straightforward as it seems.

USP focuses on building worldwide partnerships in standards-setting activities.

Increased manufacturer outsourcing requires clear policies and written agreements with CMOs.

Greater sophistication in 3D X-ray imaging technology raises its utility for QA/QC in manufacturing.

The EMA's Committee for Medicinal Products for Human Use has recommended granting of marketing authorizations for the first two monoclonal antibody biosimilars.

Cocrystals can enable the formulation of solid dosage drugs, but the FDA's final guidelines have left concerns about how their use could impact development timelines, the drug product manufacturing process, and the intellectual property position of products containing cocrystals.

The Alliance for Fair Trade with India is seeking action to resolve discriminatory trade practices and improved protection of intellectual property rights.

ISPE study reveals quality systems of manufacturing as the leading cause of drug shortages.

USP appoints regulatory experts to elemental impurities implementation advisory group.

EMA's revised guideline on biosimilars containing biotechnology-derived proteins is published for public consultation.

FDA cites cGMP violations of finished pharmaceuticals at the company's facilities in Marion North Carolina and Jayuya, Puerto Rico.

FDA Discovers Microbial Contamination in Compound Pharmacy Products

The company is cited for using unapproved visual-inspection methods for finished parenteral drugs and conducting inadequate visual inspections.
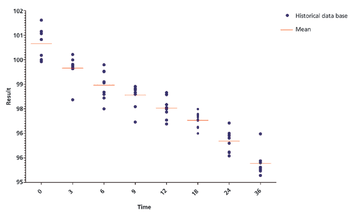
The authors discuss three methods for identification of out-of-trend (OOT) results and further compare the z-score method and the tolerance interval in OOT analysis for stability studies.