
Failure to disclose info may work sometimes, but eventually every question will be answered.

Failure to disclose info may work sometimes, but eventually every question will be answered.

The importance of new drug trials to patients, the economy, and science.

Understanding the differences between convenience, target, and self-selected samples.
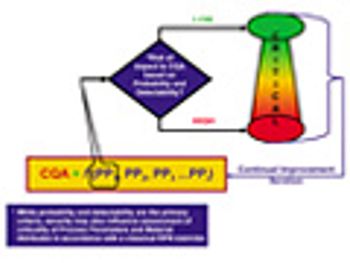
The authors provide an overview of the new ISPE Guide Series on Product Quality Lifecycle Implementation and how the guides can be used in a complementary way with existing guidance from FDA and the International Conference on Harmonization.

A Q&A with Erik van den Berg, CEO of AM-Pharma, on recent industry trends.

Soaring opioid use creates challenges for new drug development and supply-chain control.

Comparison of the top GMP deficiencies cited by the PIC/S Participating Authorities.

The confluence of science, technology, and regulation can provide path forward.

China's drug-distribution network has been a mess for years, but government reforms and industry focus are unveiling new opportunities for market order and growth.

The contract provider needs to know as much as the NDA holder.

The authors detail the possible consequences of noncompliance and a lack of quality control.

Q&A with Peter Smith and David Elder, Strategic Compliance Consulting, PAREXEL International, on acceptable deviation investigations. Smith and Elder are both former senior officials with FDA.

On Mar. 13, 2012, FDA issued a Warning Letter to Steven Victor, CEO of IntelliCell Biosciences for a violation of the Food, Drug, and Cosmetic Act, and for violations of cGMP and Good Tissue Practice in the manufacture of its adipose tissue-derived stem-cell product.

The European Medicines Agency has recommended that manufacturing processes for two anticancer medicines be transferred from Ben Venue Laboratories to alternative facilities. The decision follows GMP shortcomings identified at the US-based manufacturer in late 2011, and marks the end of the EMA's review of all fourteen centrally authorized medicines that were produced at the site.

FDA Issues Warning Letter to Warner Chilcott; Sigma-Aldrich Expands in Asia; and More.

On Mar. 12, 2012, FDA released a draft guidance document for industry direct-to-consumer television advertisements that describes FDA's plans for implementing predissemination review.

CML - Quality Assurance

FDA has issued a list of planned draft and final guidance documents for release throughout 2012. There are approximately 50 such guidances planned.

In a world where product recalls can mean the end of a company, all batches must be perfect.

Regulatory bodies, standard-setting organizations, and industry seek to tackle the problem of counterfeit drugs and securing the flow of pharma ingredients.

Brazil's generic-drug market is growing steadily.

US Pharmacopeia documents best supply-chain practices and seeks broad input on proposal.

SOCMA's Bulk Pharmaceuticals Task Force outlines key goals and challenges for user-fee legislation.

Has the long-awaited guidance answered all of the industry's questions?

Social media use raises questions about applying old standards to new information technology.