
In July 2012, new pharmacovigilance legislation will come into effect across the EU as a result of changes adopted in 2010, specifically EU Regulation No. 1235 and Directive 84.

In July 2012, new pharmacovigilance legislation will come into effect across the EU as a result of changes adopted in 2010, specifically EU Regulation No. 1235 and Directive 84.

The United States Pharmacopeia has stated that references to General Chapter <911> "Viscosity" will be changed to General Chapter <911> "Viscosity-Capillary Viscometer Methods," <912> "Rotational Rheometer Methods," or <913> "Rolling Ball Viscometer Method."

Q&A with Peter Smith, Strategic Compliance Consulting, PAREXEL International, and a former senior official with FDA, on change management best practices.

IQ Consortium representatives explore and define common industry approaches and practices for applying GMPs in early development.

BIO is calling for a more patient-centric approach to user-fee reauthorization.

US Pharmacopeia documents best supply-chain practices and seeks broad input on proposal.

New price-control policy has domestic and global firms waiting on the sidelines to launch products.

New legislation, government programs aim to bolster drug discovery and reduce regulatory hurdles.

Does global development have to entail multiple comparability studies?

Even the slightest of errors in exponential calculations can cause the biggest of headaches.

The EDQM (European Directorate for the Quality of Medicines and HealthCare) are looking to recruit a Scientific Programme Officer to join the team for a five year period to organise, manage and monitor a number of projects related to Pharmaceutical Care and Anti-Counterfeiting activities with a particular focus on the implementation of the MEDICRIME Convention

The US Pharmacopeia has issued a third round of improvements to its blood thinner standard.

ICH Q11, the anticipated guideline from the International Conference on Harmonization, titled Development and Manufacture of Drug Substances, has achieved international consensus. Q11 has been one of the fastest guidelines to move through the ICH harmonization process.

The UK's Medicines and Healthcare products Regulatory Agency has launched a new anticounterfeiting strategy with the aim of curbing the occurrence of falsified medicines in the county's supply chain.

Manufacturers seek clear path to develop safe-use approaches for more risky OTC therapies.

It's better to catch costly mistakes in the laboratory before they reach the accounting department.

The author outlines the scientific aspects of forced degradation studies that should be considered in relation to ANDA submissions.

Apple's experience with manufacturing facilities in China present opportunity for future best practice.

How niche strategies can offer mainstream potential for biopharmaceutical companies.

Poland's government aims to make the Eastern European country a biotech powerhouse.

Q&A with David Elder of Strategic Compliance Consulting, PAREXEL International on responding to a 483 within 15 days. Elder is a former senior official with FDA.
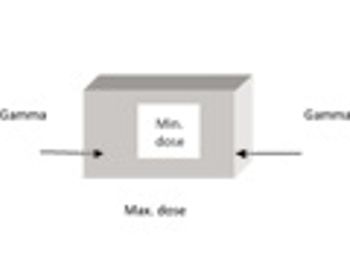
This paper examines the process of gamma irradiation of plastic materials used as part of single-use disposable systems in the pharmaceutical and biotechnology sectors, with a focus on validation requirements.

Parametric release and real-time testing use manufacturing data to ensure that products are made according to defined standards. PharmTech talks to Boehringer Ingelheim's Heribert Hausler about these issues.

The authors provide a review of test methodology and standards, including current industry and regulatory proposals, for biological indicator growout times.

FDA has issued Warning Letters to 10 companies that manufacture and distribute dietary supplements that contain dimethylamylamine. FDA cited the companies for marketing the supplements without submitting evidence that the products are safe.