
Pharmaceutical Technology Europe
... there is a huge manufacturing challenge involved in bringing cell therapies (especially stem cell therapies) from the lab bench into the clinic.

Pharmaceutical Technology Europe
... there is a huge manufacturing challenge involved in bringing cell therapies (especially stem cell therapies) from the lab bench into the clinic.

Pharmaceutical Technology Europe
So what is the EU to do? It must grant its younger researchers greater scientific autonomy and academic independence.
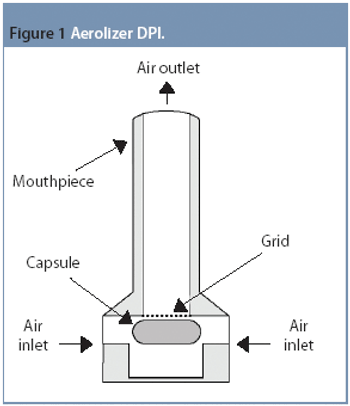
Pharmaceutical Technology Europe
There have been significant advances, especially in understanding the role of carrier properties on the aerosol performance of the API.

Pharmaceutical Technology Europe
Liability issues are often controversial, but much depends on the nature of the agreement.
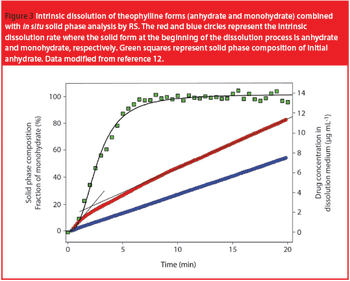
Pharmaceutical Technology Europe
When a dosage form or an API is introduced to the gastrointestinal tract, or dissolution media mimicking it, a transformation from a metastable to a more stable form with lower solubility and bioavailability is possible.
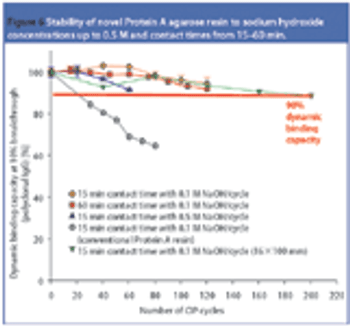
Pharmaceutical Technology Europe