Specialty Dosage Drugs
Latest News
Latest Videos
More News
Novel genetic engineering technologies are transforming the design and manufacture of bispecific antibodies, which are emerging as a promising new class of biologics.

Dr. Ulrike Lemke, Head of Sterile Fill Finish at Recipharm, sat down with Grant Playter, associate editor, to discuss drug delivery forms.
EnteroBiotix is harnessing the power of the microbiome to target an array of diseases.

TFF Pharmaceuticals announces findings that inhaled niclosamide significantly inhibits viral replication of the Omicron variant of SARS-CoV-2.

Experts share best practices in designing, manufacturing, and scaling up dry powder inhaler and metered dose inhaler drug–device combination products for inhalation drug delivery.
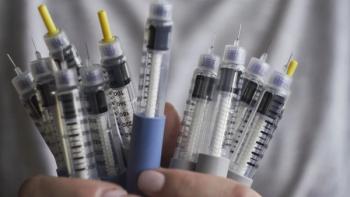
Considering interactions and understanding regulatory requirements are crucial when developing a drug or biologic product for use with a medical device.

ImmunityBio will proceed with a Phase I/II/III trial in South Africa of their dual-antigen T-cell vaccine as a boost in participants who were previously vaccinated against COVID-19.

Aprecia and Glatt investigate manufacturing technologies to widen the formulation design space for oral delivery.

The collaboration will evaluate Nanoform’s fast dissolution nanoparticles in Aprecia’s ZipDose 3DP technology platform for buccal and oral drug delivery.

Topical drugs are attractive to patients, but these complex products pose challenges for formulators.

Smart packaging opens doors to new products for diagnosis and treatment of eye problems.
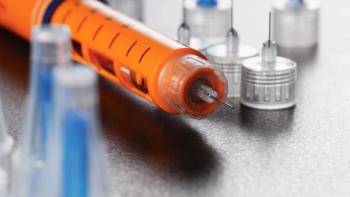
A device manufacturing process must be carefully designed in the early stages of development to ensure success in commercial manufacturing.

Inovio received a $71-million contract from the US Department of Defense to scale up manufacture of its Cellectra smart device to deliver its DNA vaccine.

New options for pharmaceutical tubes enhance functionality, sustainability, and brand identity.

Evonik opened a new facility for GMP manufacturing of bioresorbable polymers and excipients for pharmaceutical and medical applications in Birmingham, AL.
Equipment and process optimization must be considered for scaling up these developmental technologies to commercial production.

Understanding of scale-up parameters and use of process analytical technology are important to meet demand for larger batch sizes.

Trends affecting primary pharmaceutical packaging include the shift to more complex drug-device combination products.

PATH’s Microarray Patch Center of Excellence aims to accelerate transdermal patch technology for public health needs.
Several factors must be considered when reformulating APIs for pediatric, geriatric, and other specialty patient populations.

Regulatory, cultural, and technical differences between drug and device development can pose challenges to pharmaceutical combination product development.

Cure Pharmaceutical secured a patent that lets the company load higher drug amounts to its proprietary drug delivery system, CUREfilm.

Tergus Pharma announced a partnership with Great Point Partners and construction of a new commercial manufacturing facility in North Carolina.

Wearable and smart devices allow user-friendly subcutaneous drug delivery.

Virpax’s Patch-in-a-Can technology delivers pain medication using a metered-dose spray film.