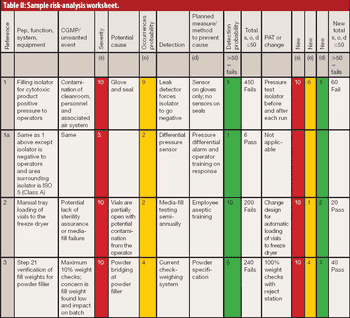
The author reviews the draft guidance on process validation, its QbD applications, and its potential impact on sterile manufacturing operations.

The author reviews the draft guidance on process validation, its QbD applications, and its potential impact on sterile manufacturing operations.
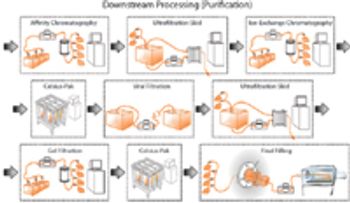
The authors discuss current and future disposable technologies and outline the validation and qualification steps that would be required for a possible disposable process stream.

The authors review the role of automation in aseptic processing and describe their experience in implementing advanced technologies, including the use of isolators and robotics.

Identification of fungi, especially filamentous fungi, has been a very difficult task. Because of the amount of experience required to accurately identify filamentous fungi to the species level, it has become acceptable to either identify these organisms to the genus level or, in some cases, simply identify them as "molds."

The author provides a history of the information chapter USP ‹1211› "Sterilization and Sterility Assurance of Compendial Articles," from the early 1900s to the current version.
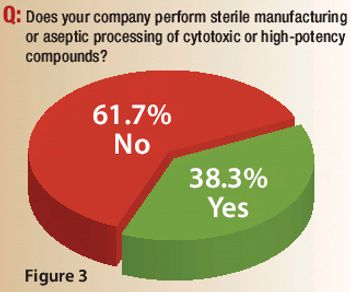
A Pharmaceutical Technology survey examines capacity expansions, outsourcing practices, innovation levels, and the role of quality by design in sterile manufacturing and aseptic processing.

The author describes various manufacturing processes and evaluates whether the guidance can be applied to each of them.