
Drug manufacturers, distributors, and dispensers oppose Buy American policies as likely to reduce reliable supplies and raise product costs.

Drug manufacturers, distributors, and dispensers oppose Buy American policies as likely to reduce reliable supplies and raise product costs.

The task force will assist in the developing, authorizing, and monitoring of treatments of COVID-19.

The guidance answers questions on changes made to the regulatory framework to address challenges during the COVID-19 pandemic.

The agency has been tasked with coordinating efforts to mitigate shortages of medicines within Europe during the COVID-19 pandemic.

The agency sent a warning letter to Genesis II Church for marketing fraudulent chlorine dioxide products the company claims treats and prevents COVID-19.

FDA is encouraging alternative insulins and challenging anticompetitive practices.

Addressing data integrity, quality culture, aging facilities, investigations/corrective actions and preventive actions, and risk management is key when conducting audits, says Susan J. Schniepp, executive vice-president of post-approval pharma and distinguished fellow, Regulatory Compliance Associates.

Because conventional cleaning methods can risk product loss, biopharmaceutical manufacturers are often reluctant to use PDE/ADE limits to validate cleaning processes.

As the coronavirus pandemic unfolds, Pharma must practice science over hype.

With ingredients sold to multiple markets, excipient manufacturers must understand the different regulatory requirements for pharma vs. food.

Pharmaceutical Technology spoke with Jens Andersson, purchasing director at Cambrex Karlskoga, about the best way to ensure the security of the bio/pharmaceutical materials supply chain.

Risk assessments, audits, and good communication between sponsor and supplier are key elements of supplier oversight.

With new tests showing NDMA levels increase under normal storage conditions, FDA calls for removal of ranitidine products from the market.

Tools help improve understanding of excipient risk in formulating OSD drugs.
Understanding European GMPs and new rules from China for excipients are crucial for formulating solid-dosage drugs.

The bill includes multiple less-noticed provisions to bolster healthcare programs and to advance the development of new treatments and preventives to combat the virus.

Noting traditional clinical trials for COVID-19 convalescent plasma will take time, FDA is allowing physicians to submit requests for single-patient emergency INDs.

FDA is offering advice and added flexibility to help sponsors adjust ongoing and planned clinical research programs during the COVID-19 outbreak.

USP technical advisors will offer assistance to drug developers to ensure material quality and testing.

The Pharmacovigilance Risk Assessment Committee (PRAC) of EMA has recommended that women stop taking 5-mg ulipristal acetate (Esmya and generic medicines) for the treatment of uterine fibroids while a safety review into potential liver injury risk is performed.

FDA postpones routine domestic facility inspections due to the COVID-19 pandemic.
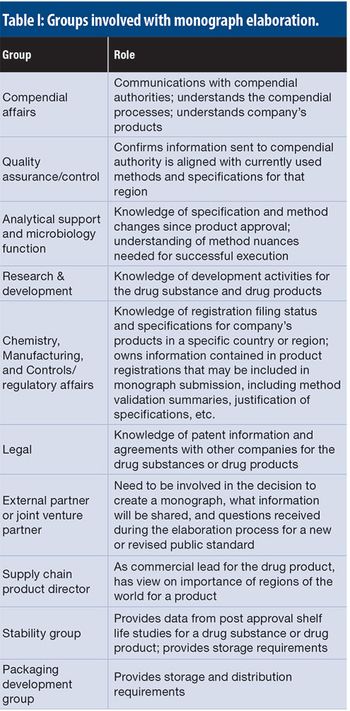
This article details the more operational aspects of monograph submissions, answering the question of how to participate.

The authors present a case study with raw materials and excipients, where a consistent, cross-functional approach is needed to ensure the appropriate selection, sourcing, testing, and filing of the materials used to manufacture bio/pharmaceutical products in a global environment, ensuring compliance with applicable compendial and regulatory requirements.

This article summarizes all the considerations that go into a company’s compendial affairs program and to look ahead at topics that will likely result in further evolution in the pharmacopoeias around the world. This look into what is on the horizon is important to help companies prepare for the inevitable changes and ensure the continued supply of quality medicines to patients globally.

This series is intended to address the challenges for the industry to comply with pharmacopoeial requirements. This article returns to this important topic with a case study at the intersection of monograph development and compliance.