
Past mistakes and misstatements have adversely influenced industry decontamination practices with vapor phase hydrogen peroxide, and this article endeavors to clarify the process.

Past mistakes and misstatements have adversely influenced industry decontamination practices with vapor phase hydrogen peroxide, and this article endeavors to clarify the process.
Regulatory mandates, niche diseases, and patient-centric solutions have all impacted pharma packaging over the years and are expected to help shape the future of the sector.

Without careful consideration and understanding, new regulations for medical devices could lead to the withdrawal of combination products from the market.

The guidance describes procedures for obtaining an additional National Drug Code for prescription drugs imported into the United States.

Glenmark Pharmaceuticals is recalling unexpired lots of Ranitidine Tablets due to potential presence of N-nitrosodimethylamine (NDMA).

FDA sent a warning letter to Dercher Enterprises, Inc., DBA Gordon Laboratories, for CGMP violations and adulterated drug products.

The European Medicines Agency and its European partners have launched a pilot program for cooperation in the inspection of facilities that manufacture sterile drug products.

The revision process and the resulting publication of proposed and official updates for pharmacopoeias around the world are described.

This article describes the revision process and the resulting publication of proposed and official updates for pharmacopoeias around the world.

An effective surveillance program for monitoring the activities of pharmacopoeias around the world requires processes, people, and tools from across a company.

An understanding of global and national pharmacopoeias is crucial to understanding change processes and access to different markets.
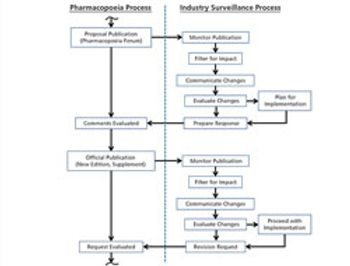
The process used to monitor and participate in pharmacopoeial changes is described.

Connect with pharmaceutical and healthcare regulatory authorities around the world via this directory.

Find links to pertinent regulatory and standard setting resources, guidance documents, and guidelines.

Long a staple for quality by design and solid dosage form manufacturing, design of experiments is becoming an integral part of biopharma upstream process development.

Warning letters tell the tale of missteps by drug companies and offer a path to compliance for quality teams that monitor these enforcement actions.

Too narrow a focus on regulatory compliance may prevent organizations from embracing-and profiting from-quality and operational excellence.

An effective surveillance program for monitoring the activities of pharmacopoeias around the world requires processes, people, and tools from across a company.

An effective surveillance program for monitoring the activities of pharmacopoeias around the world requires processes, people, and tools from across a company.

This article describes the revision process and the resulting publication of proposed and official updates for pharmacopoeias around the world.

According to a consent decree of permanent injunction issued in September 2019, the health and wellness companies must recall and stop distributing products until the companies comply with the Federal Food, Drug, and Cosmetic Act and other requirements included in the consent decree.

While labor and tariff reforms in the revised North American trade agreement may have more visible impacts on the United States economy, the final document levels a major blow to exclusivity and patent protections important to innovator biotech and pharma companies.

FDA has an official new leader, following a Senate vote to confirm Stephen Hahn as the agency’s next commissioner.

The biologic is a novel bone builder that has a dual effect of increasing bone formation and reducing bone loss.

The agency has approved three applications for generic versions of Gilenya (fingolimod), Novartis’ blockbuster multiple sclerosis drug.