
The report details OPQ’s accomplishments over the past five years.

The report details OPQ’s accomplishments over the past five years.

The research community is moving quickly to launch clinical trials of potential countermeasures, while regulatory authorities aim to support product development through regulatory flexibility.

The two agencies are collaborating to support a robust biologics marketplace by taking steps to deter anti-competitive business practices.

FDA published draft guidance for applicants seeking licensure of a proposed biosimilar or proposed interchangeable biosimilar.

Can the pharmaceutical industry embrace the goals of Quality 4.0?

The FDA Commissioner plans to address drug prices, the drug approval process, and supply chain issues during his time as commissioner of FDA.

Increased reliance on foreign producers raises concerns and spurs collaborations.

ICH will be taking industry comments under consideration when it revises its Q9 guideline in order to clarify QRM requirements, says Susan J. Schniepp, executive vice-president of post-approval pharma and distinguished fellow, Regulatory Compliance Associates.

Data management is crucial in bio/pharmaceutical laboratory settings from discovery steps through clinical studies and varies based on the development phase.

Researching excipient grades and sources, as well as screening suppliers and materials, form the basis of programs to mitigate risk.

Complex protein structures pose analytical challenges that can be addressed by advanced mass spectrometry technologies and workflows, which can be used to comprehensively characterize them.
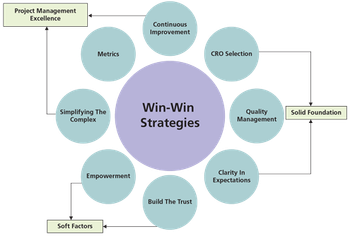
How to adopt win-win strategies and understand quality agreements for complying with cGMP when building strategic relationships with pharmaceutical contract research organizations.

Pharmaceutical Technology's In the Lab eNewsletter
The company’s new Milliflex Oasis System provides enhanced result reliability, increased productivity, and advanced traceability.

The agency has published seven guidance documents directed at the development and manufacture of gene therapies.

The agency celebrates the efforts it has made in creating a system for the evaluation and supervision of medicines throughout the European Union.

FDA revised the guidance after industry feedback and to clarify CGMP requirements for outsourcing facility operations.

The agency’s joint Big Data Task Force and the Heads of Medicines Agencies proposed actions for the use of big data to support innovation and public health.

FDA sent a warning letter to Health Pharma USA after an inspection found the company’s quality unit was not properly overseeing its drug manufacturing operations.

FDA has granted fast track designation for Novavax’s NanoFlu, a recombinant quadrivalent flu vaccine, for use by adults age 65 years and older.
Equipment and Processing Report
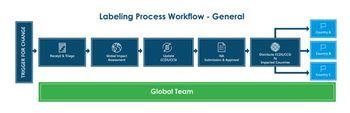
Tracking and implementing label changes are crucial to the lifecycle of a marketed drug product.

As mergers continue and operations become more complex, simplifying procedures and training can prevent costly morale, quality, and compliance problems.

Top of CDER’s to-do list for 2020 is tracking adverse events more effectively and combating the opioid crisis.

The company has various projects and launches scheduled for 2020 as part of its anniversary celebration.

The numbers of new molecular entities approved in 2019 are close to or exceed FDA’s performance in most previous years.

The agency sent a warning letter to Henan Kangdi Medical Devices Co. Ltd after an inspection found CGMP violations that included a variety of failures of the company’s quality unit.