Pharmaceutical Technology Europe
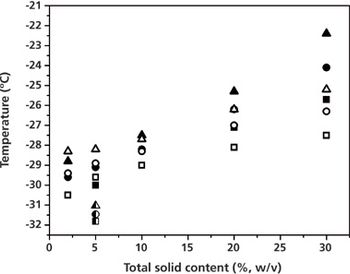
For true freeze-drying cycle optimisation, information on the maximum allowable product temperature during primary drying is required.
Henning Gieseler, PhD, works in the Department of Pharmaceutics at the University of Erlangen, Erlangen, Germany 91058. tel. +49 9131 85 29545, [email protected].

Pharmaceutical Technology Europe
For true freeze-drying cycle optimisation, information on the maximum allowable product temperature during primary drying is required.

Cycle design and robustness testing using advanced process analytical technology.
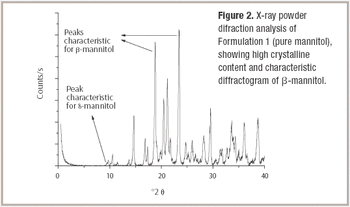
Mannitol is the most commonly used bulking agent in freeze-drying formulation design. The benefit of using mannitol is that it crystallizes during freezing and permits drying processes at higher product temperatures, and thus with higher sublimation rates relative to purely amorphous systems (1). Mannitol, however, is known to form different crystalline modifications which compromises reproducibility of product characteristics and storage stability due to phase transformations (2, 3).
October 2nd 2008