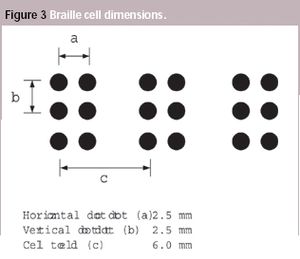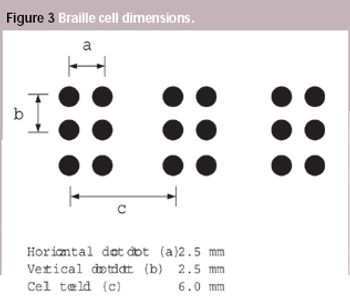
A European Commission directive requires suppliers to incorporate information on medicines in formats suitable for blind and partially sighted people, although the associated standard has not been introduced yet. This article looks at some of the issues that will need to be addressed when the standard is adopted.