Pharmaceutical Technology Europe
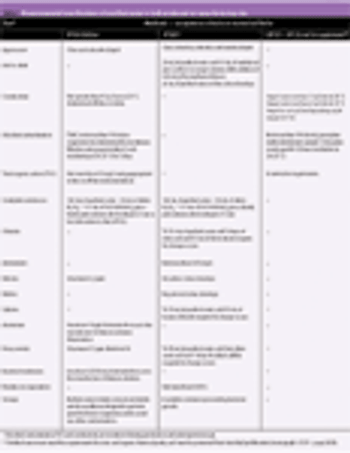
Chemical purity is the most important quality characteristic of a pharmaceutical substance. This article describes the latest scientific and technological advances to meet recent pharmacopoeial and regulatory requirements regarding the control of organic impurities in synthetically produced active substances. Future developments and suggestions for those working in quality control and raw material selection are discussed.