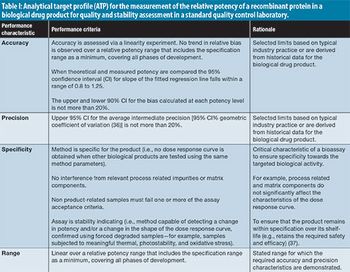
Quality-by-design risk assessment workflows can be laborious and have limited capacity to connect with vast volumes of prior knowledge generated by organizations. Their digital transformation can improve efficiency, reduce human errors, and facilitate integration within a much broader digital ecosystem.