
A judge ruled that Regeneron’s mAb be removed from the market after Amgen alleged that the marketing of Praluent hurt its reputation as innovator of this class of drugs.
Randi Hernandez was science editor at Pharmaceutical Technology from September 2014 to May 2017.

A judge ruled that Regeneron’s mAb be removed from the market after Amgen alleged that the marketing of Praluent hurt its reputation as innovator of this class of drugs.

In an attempt to battle low production volumes of hard-to-manufacture biopharmaceuticals, GE Healthcare announced on January 4, 2017 that it will partner with Synpromics on the development of synthetic promotors. The goal of the collaboration is to identify promoters that will work most effectively with GE’s existing expression system for optimal transcription.

FDA’s Center for Drug Evaluation and Research (CDER) approved 22 new molecular entities (NMEs) in 2016, according to a January 4, 2016 release from the agency. Of the 22 NMEs, 12 were large-molecule therapeutics. Of CDER’s 45 novel drug approvals in 2015, 17 were considered large-molecule therapeutics (larger than 900 Daltons).In 2016, 11 of the 12 large-molecule drugs that were approved were biologics. This includes seven monoclonal antibodies (mAbs), one hormone, and three DNA-derived medications. The remaining large-molecule medication was a diagnostic agent.
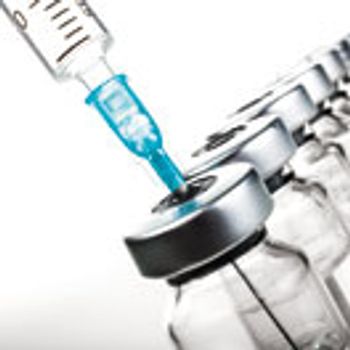
Improvements to aseptic manufacturing procedures are long overdue. But how feasible is it for manufacturers to modernize fill lines of legacy products?

Amgen discussed the drug-delivery approaches to two of its biologics in a recent second-quarter earnings call.

The move follows a warning from FDA wherein FDA cited ALK-Abello with numerous manufacturing violations.

The Phase I trial will test Bavarian Nordic’s vaccine, which is manufactured using a vaccine vector based on smallpox.The recent resurgence of yellow fever incidences over the past six months has prompted health officials to ramp up the fight against the virus. Like the Zika virus, yellow fever is transmitted primarily through the bite of infected female Aedes aegypti mosquitoes.

The therapeutic candidate AZD8601is an investigational mRNA-based therapy that will be tested for its ability to regulate the protein that influences vascular tissue growth.

The vaccine candidate has also won a Priority Medicines (PRIME) status from EMA.

Amgen and Allergan’s biosimilar to popular breast cancer drug Herceptin is comparable to the innovator molecule, Amgen recently announced.

A new website collects and rates information about life-science supplies from academic journals to simplify researcher purchasing decisions.

Thirty percent of current Remicade patients could be eligible for a switch to competitors such as Inflectra, according to estimates, although J&J doubts this will occur in the United States in 2016.

The collaboration will focus on the investigational candidate JTX-2011 and up to four other early-stage programs in immune-oncology.

A new study examines the oral delivery of protein drugs in plant cells and hypothesizes that the cold storage and transportation of biologics could someday be eliminated.

FDA reviewers have found that Sandoz's GP2015, a biosimilar version of Amgen's Enbrel, is highly similar to the original product in terms of purity, safety, and potency.

The agency says that the routine large-scale compounding of drugs that are exact copies of existing medications undermines the the drug approval process.

Antibodies to dengue may either confer protective immunity to the Zika virus or enhance disease severity in secondary, related infections such as Zika.

A research team associated with Dr. Carl June announces it has discovered a way to engineer a patient’s own immune cells to recognize cancer-specific glycoantigens on tumor cells.

In a lawsuit against Celltrion, Janssen alleges the GE subsidiary infringed one of Janssen’s patents covering the method of making the cell-culture media that HyClone later supplied to Celltrion.

A new indication for Emergent BioSolutions’ BioThrax will give the drug market exclusivity through November 2022.

Results of a Phase II clinical trial reveal that stem-cell transplantation treatment following complete immune system destruction increases the duration of long-term remission in patients with multiple sclerosis.

The inactivated vaccine, manufactured with a GE FlexFactory system, could be associated with fewer adverse reactions than the live vaccine option.

Uncertainty about the demand for a biologic medication can be partly mollified with some well-planned capacity outsourcing, contends a new report by ORC International sponsored by Patheon.

An article in Nature Reviews Drug Delivery reports the clinical success rates of biologics, and these numbers differ from those recently highlighted in a similar BIO study.

Results of the HERITAGE study were announced at the 2016 American Society of Clinical Oncology meeting.

New pharmacovigilance activities seek to support the creation of guidelines for safe switching from innovator products to biosimilars.

As the battle for ownership over the use of CRISPR to edit DNA heats up, some of the CRISPR pioneers refocus their efforts to demonstrate the technology’s applications for editing RNA.

PharmTech sat down with an intellectual property lawyer to examine how companies are protected when they engage in activities where sharing of trade secrets must occur.

Samsung Bioepis and partner Biogen announced on May 30 that the European Commission approved Flixabi for the treatment of six inflammatory conditions.

In the largest-ever report of its kind, drugs entering clinical development in Phase I were found to have only a 1 in 10 chance of FDA approval.