Pharmaceutical Technology Europe
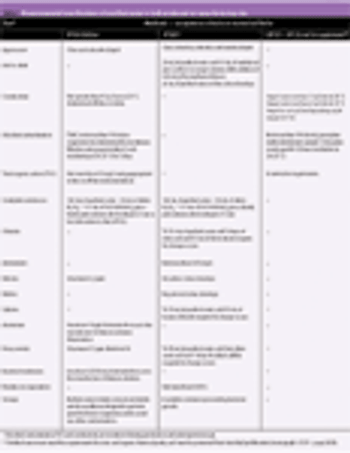
Size reduction of materials through comminution is employed in many industries, including agrochemicals, minerals, ceramics and pharmaceuticals. The reasons for particle size reduction depend on the industry in question. Within the pharmaceutical industry, a large percentage of products are formed from powders and undergo processing to improve dosage form properties. Particle size reduction prior to compacting to tablets can aid with dissolution and homogeneity. Such processing of powders is, in part, dependent on their mechanical properties and balancing these properties is crucial in achieving desired manufacturing performance. Generally, pilot-scale milling trials are run to determine the most effective and efficient mill and operating conditions for each material. These trials, however, require relatively large quantities of material as well as time, and are normally run in early development when sufficient material becomes available. Hence, it would be highly beneficial to identify a physical property..