Increasing potency and growing interest in antibody-drug conjugates are creating challenges for manufacturers of HPAPIs.

Increasing potency and growing interest in antibody-drug conjugates are creating challenges for manufacturers of HPAPIs.

The international pharmaceutical industry is changing its approach to R&D and is increasingly relying on outsourcing for drug discovery.

SyncOperations Software Helps Increase Workflow

A new process analytical technology based on impedance spectroscopy has potential applications for characterizing product attributes during the freeze-drying process.

Encapsulation technologies for solubility/bioavailability enhancement, controlled release, and formulation of drug combinations and patient-friendly dosage forms.
USP evaluates raw materials used in the chemical synthesis of peptides.

Kurt Lumsden, Director, eCDS Client Services at PAREXEL Informatics, discusses eClinical supply-chain management.

Mark D. Kramer talks about combination products from a regulatory perspective and explains the implications of FDA's final rule on current GMP requirements for combination products.

Higher antibody titers and a growing demand for smaller-volume, flexible processes are creating the need for more cost-effective downstream processing.

More media options open publishing opportunities for drug development experts.

How badly is Brazil's pharmaceutical market suffering from the global instability of emerging markets?

Graham Reynolds of West Pharmaceutical discusses glass versus plastic packaging.

New identifiers and tracking requirements aim to block illegitimate products.
Methods of validating and assuring container closure integrity are crucial for quality assurance.

Arc View Handheld Mobile Sensor Enhances Measurements

CLARiSUITE Software Improves Accessibility and Productivity

Multirobot Offline Programming Software Allows 3-D Simulation

Outsourcing activity remains strong and unlikely to abate, especially in more traditional areas.

As Europe strives to firmly incorporate quality-by-design principles, there are several key issues that still need to be addressed.
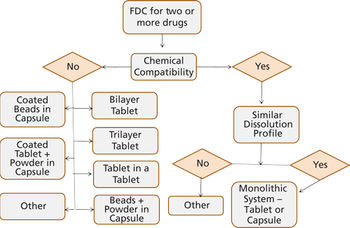
Anil Kane from Patheon spoke to Pharmaceutical Technology about the challenges in the development of FDCs and discussed how to integrate quality-by-design (QbD) in the manufacturing process of FDCs.

Click the title above to open the Pharmaceutical Technology April 2014 issue in an interactive PDF format.