
Ensuring compliance though increased statistical knowledge and resources.

Ensuring compliance though increased statistical knowledge and resources.

Can microdosing make medicines safer and more effective for children?
More sophisticated biological expression systems expand the functionality of the traditional systems for protein synthesis.
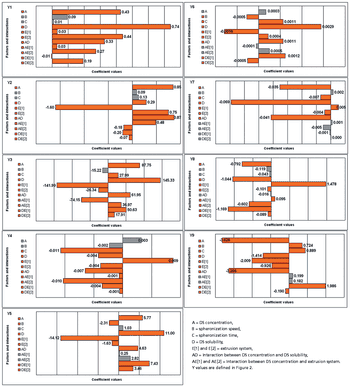
The authors compare three systems of single-screw extrusion using binary formulations for their suitability for producing pellets of various formulations and under various spheronization conditions.

Why SOPs are rarely followed, often cited, and in need of follow-through.

Academic–industry partnerships are increasingly important in biopharmaceutical innovation.

Editor's picks of new manufacturing products for June 2011.

A Focus on Creative Strategies to Help the Developing World.

A recent industry survey shows keen interest in improving bioreactors and cell-culture media.
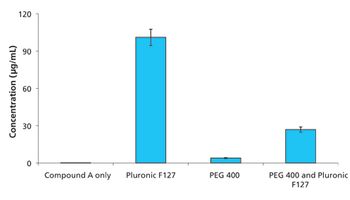
The study results suggested that Pluronic F127 might be a potent inhibitor of drug precipitation for Labrasol formulations.

Industry struggles to curb drug abuse, diversion, and disruptions in supply.

Sometimes, there are just too many cooks in the kitchen.
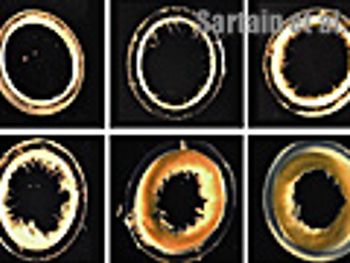
A holistic approach to establishing robust control measures.

FDA weighs multiple views regarding the Biologics Price Competition and Innovation Act.
Industry associations will soon provide new recommendations about extractables and leachables.

Christopher Meissner, CEO of Meissner Filtration Products, on recent industry trends.

This month's BIO Convention will encourage needed conversations.

India's drug pricing authority increased the retail costs of certain domestically manufactured drugs, but not those of imported drug products. Pharma is asking why.

Effective containment in API and drug-product manufacturing encompasses a variety of process, equipment, and operational issues.

Next-Gen Expression Systems