
ePT--the Electronic Newsletter of Pharmaceutical Technology
Aptuit, Actavis, BASF, Bristol-Myers Squibb, more

ePT--the Electronic Newsletter of Pharmaceutical Technology
Aptuit, Actavis, BASF, Bristol-Myers Squibb, more

ePT--the Electronic Newsletter of Pharmaceutical Technology
Basel, Switzerland (May 11)-Roche opened a new biotechnology production center in Basel today, just two years after beginning construction. The facility will manufacture "Avastin," a cancer drug that inhibits the formation of new tumor blood vessels.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Rockville, MD (May)-The US Food and Drug Administration issued a draft guidance, "Labeling for Human Prescription Drugs-Determining Established Pharmacologic Class for Use in the Highlights of Prescribing Information."

ePT--the Electronic Newsletter of Pharmaceutical Technology
Darmstadt, Germany (May 13)-Mylan Laboratories Inc. and Merck KGaA have signed a share purchase agreement under which Mylan will acquire all of Merck's worldwide operations within Merck Generics, the company?s generics business.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Washington, DC (May 3)-The Biotechnology Industry Organization published a position paper stating that any legislation establishing a regulatory pathway for follow-on biologics should grant pioneering products 14 years of data exclusivity.

ePT--the Electronic Newsletter of Pharmaceutical Technology
London (May 1)-The European Medicines Agency launched a database designed to facilitate the exchange of information about compliance with good manufacturing practices.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Boston, MA (May 8)-The global biotechnology industry showed several positive signs in 2006, including increases in overall revenues and financing, although the industry as a whole continues to operate at a loss, according to Ernst & Young's annual analysis of the biotechnology industry.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Rockville, MD (May 1)-The US Food and Drug Administration issued a report, "Critical Path Opportunities for Generic Drugs," to identify the scientific challenges, including those in manufacturing science, in developing generic drugs and the opportunities for collaborative solutions in resolving those challenges.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Dublin, OH (May 3)-In preparation for California?s new pedigree legislation, Cardinal Health plans to integrate radio frequency identification (RFID) technology into the operations at its pharmaceutical distribution center in Sacramento by fall 2007.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Althea, AstraZeneca, Bosch, more

ePT--the Electronic Newsletter of Pharmaceutical Technology
Hamilton, OH (May 2)-Amylin Ohio LLC, a subsidiary of Amylin Pharmaceuticals, is expanding construction at its manufacturing facility in West Chester, Ohio. The expansion increases the company?s total investment to approximately $400 million.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Washington, DC (April 30)-Less than two weeks after the Senate Health, Education, Labor, and Pensions Committee voted to reauthorize the Prescription Drug User Fee Act (PDUFA), the bill this week moved onto the Senate floor.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Geneva, Switzerland (Apr. 27)-A meeting of the World Health Organization and the Committee for Medicinal Products for Human Use's approval of Novartis's new cell culture-derived influenza vaccine offered new hope that sufficient numbers of vaccines could be produced in case of a pandemic.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Devens, MA, Montes Claros, Brazil, and Florence, SC (May 2)-Bristol-Myers Squibb Company, Novo Nordisk A/S, and Roche provided updates to their companies? respective manufacturing expansions.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Rockville, MD (May 1)-The US Food and Drug Administration issued a guidance to alert pharmaceutical manufacturers, pharmacy compounders, repackers, and suppliers to the potential public health hazard of glycerin contaminated with diethylene glycol (DEG), a poison.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Albany Molecular Research, BAC, Crucell, More

Breaking up is easy to do.

USP exercises a vigorous standards-setting process that draws on both public comments and meticulous scientific review.

A recent benchmarking report on pharmaceutical manufacturing raises questions about the performance of contract manufacturers, but further analysis also raises concerns about the process and data used to arrive at this conclusion.

Sanofi to Close Irish Plant, FDA Inspections and Warning Letters Continue to Decline, FDA Submits Final Proposals for PDUFA IV, and more.
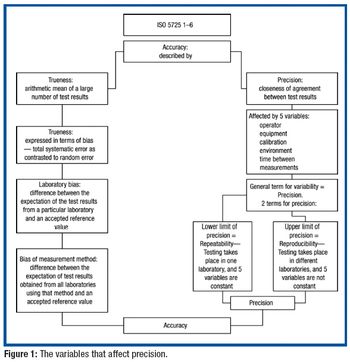
USP applies metrological principles to the dissolution procedure alone and in collaborative studies to understand and minimize potential sources of variability.

PTSM: Pharmaceutical Technology Sourcing and Management
Ricerca Biosciences LLC a contract drug-development company, is embarking on a growth strategy upon which it hopes to double its revenues by 2009. The company recently completed a financial restructuring that includes new equity and term facilities totaling nearly $50 million. That financing will enable Ricerca to implement a multiyear business plan, which includes a $15-million investment to add chemistry and biological laboratories to its site in Concord, Ohio.

PTSM: Pharmaceutical Technology Sourcing and Management
A roundup of company moves and positioning from the pharmaceutical industry and biotechnology industries and contract service providers.

BioConvergence, PharmaFab, Johnson & Johnson, More

San Juan, PR (Apr. 25)-Regulatory and quality issues were prominent discussion topics at this year?s ExcipientFest conference and exhibition.

Basel, Switzerland (Apr. 26)-Roche outlined advances it is production strategy for "Tamiflu" (oseltamivir) for treating the H5N1 avian influenza virus.

London (Apr. 23)-AstraZeneca PLC agreed to acquire MedImmune, Inc. for approximately $15.6 billion. The deal is expected to close in June 2007.

New York (Apr. 26)-Bristol-Myers Squibb Company and Pfizer, Inc. entered into a collaborative agreement for the development and commercialization of apixaban, an anticoagulant that BMS discovered.

Interphex2007, New York, NY (Apr. 26)-Robustness studies for analytical methods are critical in being able to provide the assurance to the quality of an analytical method, a topic addressed in a conference session, "Performing Analytical Method Validation Robustness for Regulatory Compliance," at Interphex on Thursday.

Interphex2007, New York, NY (Apr. 26)-Maintaining reliable equipment in pharmaceutical manufacturing operations is critical not only for achieving productivity goals and safety, it also is a prerequiste for moving operations to a lean-manufacturing environment.