
Clamor mounts over compromised care and rising costs due to lack of crucial therapies.

Clamor mounts over compromised care and rising costs due to lack of crucial therapies.

Careful mixing during a product's distillation can help avert trouble from a strong concoction.

Novartis AG and Novartis Pharmaceuticals Corporation have been focusing research efforts on rare diseases since the company was established in 1996.
The study evaluates near-infrared analysis of tablets nominally containing 4 mg of chlorpheniramine maleate and 10 mg of phenylephrine hydrochloride per dose.
The authors describe the operational qualification of test accuracy with regard to temperature drift using a thermal-compensation algorithm on several freeze dryers.
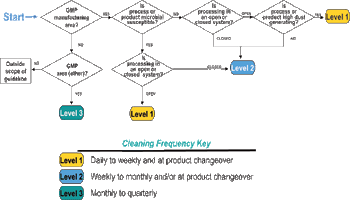
The fifth in a series of eight case studies from the Product Quality Research Institute focuses on nonsterile facility cleaning requirements.

Russia is aiming to provide an alternative to China and India for drug manufacturing, including APIs.

The authors propose a plan to keep the US pharma industry afloat and in the lead.
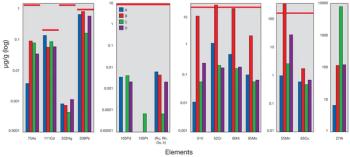
The US Pharmacopeia (USP) proposes to lower the maximum permissible limits of trace elements in pharmaceuticals and recommends that impurities be measured through automated instrumentation-based methods. The proposed regulations specify inductively coupled plasma–mass spectrometry (ICP–MS) and inductively coupled plasma–optical emission spectrometry (ICP–OES) as the techniques of choice. This article discusses the benefits of ICP–MS and ICP–OES for the accurate detection of trace elements in pharmaceutical products, in compliance with the proposed USP chapters.

Pharmaceutical Technology Europe
June and July saw three major US conferences on implementing single-use technologies: the IBC Single-use Applications meeting, the PDA Single-use Workshop and the Bio-Process Systems Alliance (BPSA) International Single-use Summit (ISUS). Jerold Martin highlights some of the key topics discussed at these meetings.
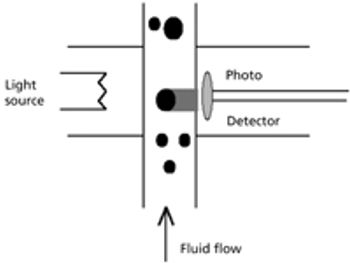
This article focuses on the history of glass delamination and methods that detect it, both from a compendial and a research perspective.

Pharmaceutical Technology Europe
It is very difficult to measure the problem of counterfeiting accurately from year to year; by definition criminals don?t file tax returns or publish quarterly earnings.

Pharmaceutical Technology Europe
In an age dominated by the internet and uncertainty over the best packaging security methods to employ, counterfeit medicines have the ideal environment to thrive.

Last week, FDA published a final guidance for pharmaceutical manufacturers that plan to incorporate physical–chemical identifiers in solid oral dosage forms as an anticounterfeiting strategy.

Equipment and Processing Report
Recent recalls have contributed to the pharmaceutical industry?s heightened awareness of glass delamination (i.e., the formation of glass flakes in a vial), which could affect drug quality and patient safety. To confront this growing problem effectively, drugmakers must understand its causes.

FDA, CMS, Medicaid Launch Pilot Program to Conduct Parallel Reviews of Medical Devices.

FDA may consider whether to add specific search criteria (e.g., those that would enable product-specific or violation-specific searches) to its inspections database to make it user-friendly and improve data analysis. The idea was included in a list of eight draft proposals for increasing transparency that the agency published on Oct. 3, 2011.

Europe establishes new collaborative system to track products.

AAPS Global Health Focus Group's Kishor M. Wasan discusses new initiatives.
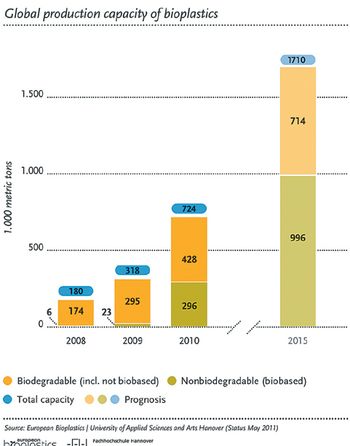
Drugmakers and packagers are pursuing various initiatives to reduce their carbon footprints. This article contains bonus material.

Getting the most value out of M&As requires proper upfront legwork.

Corporate management must be held accountable for quality at all levels.

Expanding the organization's mandate will strengthen inspections.

The authors examine risk management relating to the quality issues of clinical-trial materials and discuss areas that would benefit from additional consideration and harmonization.

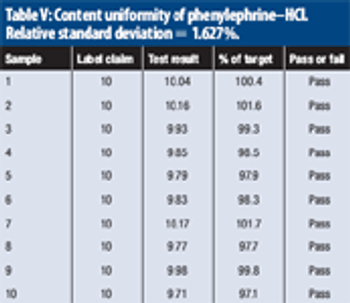
The authors describe the concept of the limiting discriminatory the limiting discriminatory threshold (LDT) as an objective means of evaluating the inherent quality requirement of a large-sample content-uniformity test.