
To keep moving forward, the Pharmacopoeial Discussion Group needs industry participation.

To keep moving forward, the Pharmacopoeial Discussion Group needs industry participation.

The European Union market takes steps toward continuous processing and modular facilities.

Copay coupons may help patients and drugmakers, but who ends up holding the bag?

The author describes how providing appropriate information about the API in the Common Technical Document can aid FDA's review of an abbreviated new drug application.

A Q&A with Gilles Cottier, president of SAFC, on recent industry trends.

New product reviews for January 2012.

Pharmaceutical Technology Europe
Multilayer tablets can be challenging, but meeting regulatory expectations is crucial. We speak with Vilayat Sayeed from the FDA, to find out more about the critical factors that need to be fully understood.

Equipment and Processing Report
Risk assessment is not a new concept to the pharmaceutical industry, but lately the phrase has become a mantra. A systemic, science-based way to manage risk is becoming essential to meeting the spirit and letter of FDA requirements.

In a Warning Letter, FDA cited "significant violations" of CGMP regulations, including several repeat observations, at three Novartis facilities located in Colorado, North Carolina, and Canada.

The FDA and EMA are moving from "confidence-building to reliance upon" each other in a step-up in cooperation on GMP inspections; the latest move following successful completion of pilot projects this summer.

Cultivating a productive investment environment will require partnerships with a range of stakeholders.

Cleanliness is crucial, even if zapping and trapping is necessary to reduce product contamination.

The seventh in a series of eight case studies from the Product Quality Research Institute focuses on packout remedies.

Added responsibilities and outside concerns prompt overhaul of agency's structure.

Contract organizations must have highly organized teams and plans to accommodate today's audits.

Drug shortages, supply-chain security, generic-drug incursion, and flexible manufacturing models are some of the issues shaping the bio/pharma industry.

Precedents set in the historic Barr case continue to raise questions over suitable sample-size criteria.

Political leaders need to consider the impact of the biopharmaceutical industry on the economy.

Packaging is indeed headed to be a lead sector in the Asian pharmaceutical environment, but certain challenges must first be overcome.

A Q&A with Deborah Tanner, executive vice-president and group president of R&D laboratories at Covance, on recent industry trends.

Readers react to the economic turmoil of the past year and look longingly forward to 2012.

Employee training-at all levels-is crucial for moving forward with a successful risk- and quality-based manufacturing strategy.

The last in a series of eight case studies from the Product Quality Research Institute focuses on internal GMP audits.
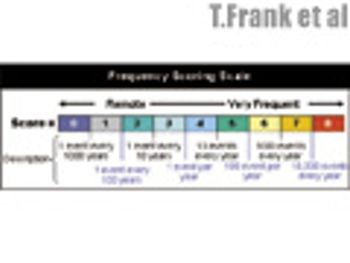
The sixth in a series of eight case studies from the Product Quality Research Institute focuses on packaging line GMP optimization.

EMA Hosts Subgroup Analysis Workshop.