
A public consultation has opened in Europe for the revised guideline on the good distribution practice of medicinal products for human use.

A public consultation has opened in Europe for the revised guideline on the good distribution practice of medicinal products for human use.

FDA issued a draft guidance for industry comment on proposed policy for diagnostic tests used with targeted drug therapies, that is personalized medicine.

FDA Approves the Influenza Vaccine Formulation for the 2011-2012 Flu Season.

The pharmaceutical industry and US regulatory bodies have not responded adequately to the increasing level of outsourced manufacturing in countries such as China and India, according to a new white paper by the PEW Health Group.

Equipment and Processing Report
Even though manufacturers are responsible for ensuring the quality of combination products, some companies may not be certain about what quality system to apply to their production.
![Rivera-Fig-4-new[2]web-731308-1408618780125.jpg](https://cdn.sanity.io/images/0vv8moc6/pharmtech/c5b3bd34e31edbde4e6a56be25a83c3d7a998f32-504x275.jpg?w=350&fit=crop&auto=format)
Equipment and Processing Report
The author describes the components that make up a clean-in-place system and how the system should be built to ensure efficiency.

Equipment and Processing Report
We?re attempting to validate a clean-in-place process using total organic carbon (TOC) as the analytical method. Our swab-sample recovery tests are giving us inconsistent results. Recovery values range from 95% to 205%, and there seems to be no correlation with the amount of drug product deposited on the coupon. What could be the problem?

FDA banned the importation of products manufactured at the Mexican unit of Dr. Reddy's Laboratories. The import ban is a result of the company's failure to correct the violations listed in a recent Warning Letter to the agency's satisfaction.

FDA issued a draft guidance, entitled "Q11 Development and Manufacture of Drug Substances," which is now available for comment according to a notice published on June 29, 2011, in the Federal Register.

The European Medicines Agency has responded positively to a new directive published in the Official Journal of the European Union addressing concerns over increases in falsified medicines in the supply chain.

An FDA panel has voted unanimously to withdraw approval for Roche's Avastin for use in the treatment of metastatic breast cancer because there is not enough data to support that the benefits outweigh the risks.

A PQRI expert working group provides case study examples of risk-management applications.

FDA, in cooperation with IPEC, is building a spectral library of excipients to detect improper ingredients within a drug product on site.

Getting an answer is easy-asking the right question is apparently more difficult.

Member states in the EU are working to implement the newly passed Falsified Medicines Directive.

Follow-on versions of complex biologics require extensive expertise in development and regulatory procedures.

US support of regenerative medicine is essential for maintaining a lead on healthcare innovation.
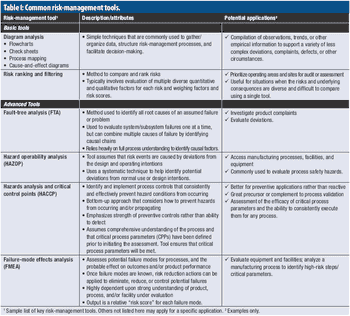
A PQRI expert working group provides case study examples of risk-management applications.

Which route will we take to arrive at a national stem-cell policy?

FDA Publishes Final Guidance on Dissolution Testing.

President Obama unveiled an Advanced Manufacturing Partnership designed to reinvigorate the country's manufacturing sector.

The European Medicines Agency (EMA) has held a second forum regarding the implementation of new pharmacovigilance legislation, which gave stakeholders the opportunity to discuss their expectations on various aspects of the new legislation's execution.

The Association of the British Pharmaceutical Industry (ABPI) has published a guidance that suggests best practices for managing adverse events and other pharmacovigilance data from the internet and social media tools.

FDA Issues Warning Letter to Dr. Reddy's Following Inspection of the Company's Mexico-Based API Manufacturing Plant.

The traditional method of conveying information in the brief summary of a printed prescription-drug advertisement is neither the most comprehensible nor the most preferred by consumers, according to an FDA study.