
The author examines sample-preparations methods used in inductively coupled plasma–optimal emission spectroscopy for four test metals.

The author examines sample-preparations methods used in inductively coupled plasma–optimal emission spectroscopy for four test metals.

Last week, Ben Venue Laboratories decided to exit the contract-manufacturing business during the next several years, thus ending more than 70 years of service in this field. To ensure the supply of medically necessary products, the company will work with its customers to develop and execute long-term transition plans.

FDA, Arkansas Sign Agreement to Enhance Regulatory Science.

Since China's State Food and Drug Administration revised its GMPs last year, the agency has been making other improvements as well to improve the quality of its pharmaceutical manufacturing industry.

Equipment and Processing Report
We try to use previous experience to develop the optimal granulation process for each new ingredient, but we sometimes get it wrong. Many times, we?ve taken materials from the granulator only to find that they exhibit poor flow and insufficient density. How can we easily determine the end point of a given granulation process?

Equipment and Processing Report
Achieving a consistent level of quality control could greatly reduce waste and save money for the pharmaceutical industry. But why has talk about Six Sigma died down at a time when it could be of great benefit?

FDA Issues Final Guidance Regarding cGMPs for PET Drugs.

EMA has concluded in a recent report that a pilot program investigating the mutual benefits of joint international inspections of API manufacturing facilities has been a success. FDA has also reached a similar conclusion based on the findings.

The Society for Chemical Manufacturers & Affiliates commented on EPA's modifications to the Inventory Update Reporting rule, also broadly know as the Chemical Data Reporting rule. EPA issued the final rule earlier this month.

Since the passage of the BPCI Act in 2009, manufacturers have been waiting for guidance from FDA on what that approval process will look like.

The International Society for Pharmaceutical Engineering published a guidance document that defines current best practices in pharmaceutical manufacturing applications for handling gases that come into direct contact with the biopharmaceutical and pharmaceutical process steams.

FDA Website Provides Information on Drug Shortages.

Rising imports and overseas production spur realignment of enforcement.

Contract manufacturing organizations throughout Asia are increasing their capabilities to meet market demand and attract foreign investment and partnerships.

Are biosimilars the next big thing or just the next big bubble?

A firm grasp of probability and ongoing re-evaluation are key.

A book about developing quality-control training manuals provides a wealth of information and a dearth of practical help.
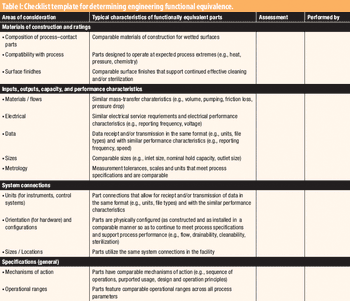
The second in a series of eight case studies from the Product Quality Research Institute focuses on functional equivalence for equipment.

Visiting a new site or going down memory lane may not get you where you want to go.

Many child-safe package designs help improve compliance and provide tamper evidence.

Terry Novak, president of Norwich Pharmaceuticals, on recent industry trends.

Quality management requires more effort in a complex supply chain.

Thomas P. Layloff describes the advantages of using thin-layer chromatography methods for counterfeit detection. This article contains bonus online material.

USP promotes horizontal standards for biologics' quality attributes.

Pfizer's Experience with QbD. This article is part of a special issue on Outsourcing.