
FDA impersonators and counterfeit drugs threaten the public's trust in online pharmacies.

FDA impersonators and counterfeit drugs threaten the public's trust in online pharmacies.

Vaccine R&D is surging, but continues to raise manufacturing and regulatory challenges.

Regulators and industry move to require inspections of API manufacturing facilities.
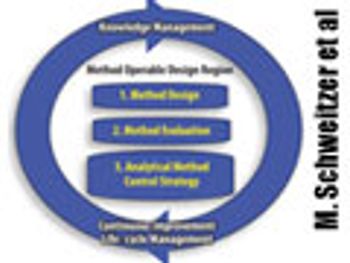
The authors present two concepts to improve robustness and facilitate continuous improvement in analytical methods. This article contain bonus online material.

Leading experts share insight on the current and future direction of process analytical technology. This article contains bonus online material.
We never thought implementing complex changes could become more cumbersome.

Pharmaceutical Technology Europe
The authors present two concepts to improve robustness and facilitate continuous improvement in analytical methods.

Pharmaceutical Technology Europe
Although representing a huge market, the EU also presents problems that hamper profitability- particularly when it comes to differing price regulations across member states.

Pharmaceutical Technology Europe
Pharma faces imminent expiries of key patents and increasing competition from generics. To protect what they already have, or what they will invent or acquire, companies must give patent protection the attention it deserves.

Pharmaceutical Technology Europe
References for the article published in the February issue of Pharmaceutical Technology Europe.

ePT--the Electronic Newsletter of Pharmaceutical Technology
New York Governor Wants Stricter Limits On Pharmaceutical Marketing; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Two US senators have introduced the Nanotechnology Safety Act of 2010, which would establish a program within the US Food and Drug Administration to assess the health and safety implications of nanotechnology in everyday products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Keiji Fukuda, special adviser on pandemic influenza to the World Health Organization's (WHO) director-general, told a hearing at the Council of Europe that WHO's response to the influenza (H1N1) pandemic was not improperly influenced by the pharmaceutical industry.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency has launched a public consultation on its Road Map to 2015, a strategic vision that lays out the agency's priorities for the next five years.

ePT--the Electronic Newsletter of Pharmaceutical Technology
FDA Issues Warning Letter To McNeil Healthcare; Charles River Laboratories Will Suspend Operations At Massachusetts Facility.

ePT--the Electronic Newsletter of Pharmaceutical Technology
McNeil Consumer Healthcare is voluntarily recalling certain lots of over-the-counter products in the Americas, the United Arab Emirates, and Fiji.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The United States Pharmacopeial Convention is recalling USP 33?NF 28.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Commission has asked certain pharmaceutical companies to submit copies of their patent-settlement agreements.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration released a warning to the public on Dec. 29, 2009 about criminals posing as FDA special agents and other law enforcement personnel as part of an international extortion scam.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Bioject Medical Technologies establishes alliance with MPI Research; Biogen Idec CEO to retire; And More.

Grace McNally, in the Office of Compliance at FDA's CDER, discusses the role of QbD and pharmaceutical quality systems as it relates to contract manufacturing.

Team leaders in FDA's Office of Generic Drugs provide an overview.

Plus, novel dosage forms and emerging trends.

Directors and staff miss the mark when it comes to following procedures.

Pharmaceutical Technology Europe
This month sees Europe make its move towards electronic submissions harmonization, but is it ready?