
ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA issues several recent enforcement letters to Cambrex, Johnson & Johnson, and Pedinol.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA issues several recent enforcement letters to Cambrex, Johnson & Johnson, and Pedinol.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Later this month, Dr. Barbara Jentges, the managing director at PhACT GmbH, a regulatory consulting and training firm based in Duggingen, Switzerland, will be speaking at the 2nd Vetter Drug Management Leadership Conference in Germany.

Thanks to their keen observations, these auditors reveal the true culprits of deviations.

After years of promomting QbD concepts, FDA's ready to take action on nonconformers.

Robotic systems provide flexibility and efficiency (and they're not as difficult to use as you think). This article contains bonus online-exclusive material.

Authenticating tools help identify counterfeit drug products. This article contains bonus online-exclusive material.
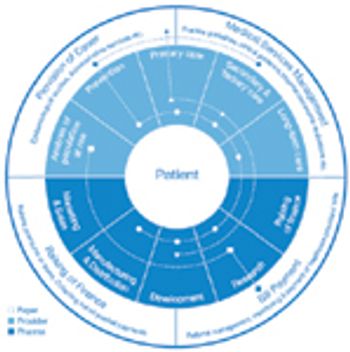
Personalized medicine and integrated healthcare delivery require new business and pricing models. This article contains bonus online-exclusive material.

Health crises generate support for new vaccines and treatments for diseases found in developing nations.

Representatives of Japan's MHLW report on recent ICH activities and what the ministry expects from Q11.

As new process validation guidelines emerge, industry needs to reinvent how it releases product.

IPEC's new stability testing guide takes into account the full supply chain's storage conditions.

Designated as a "pharmerging market," Brazil is revamping its pricing models.

The author describes the approach taken to develop a facility dedicated to handling potent and cytotoxic drug substances.
A Project Manager's Perspective.
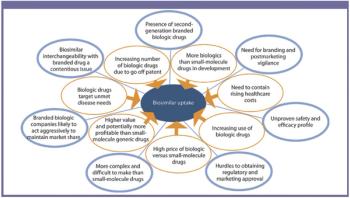
The author reviews the major biopharmaceutical markets' activity and predicts how the markets may evolve.
Pharmaceutical Technology Europe
Human embryonic stem cells are of immense interest to researchers because of their ability to potentially develop into any kind of tissue.

Pharmaceutical Technology Europe
When validating automated systems from third-party providers, using the V model and failure modes effects and criticality analysis (FMECA) early in the process can help.

The FDA has said it would like to amend postmarket safety reporting regulations ?to require that manufacturers and other facilities subject to current reporting requirements submit their reports in an electronic format".

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration announced in an Aug. 20, 2009 release that it would like to amend postmarket safety reporting regulations "to require that manufacturers and other facilities subject to current reporting requirements submit their reports in an electronic format."

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: UCB and Novartis form agreement; AAIPharma appoints VP of regulatory affairs; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, USP signed a Memorandum of Understanding with the Permanent Commission of the Pharmacopeia of the United Mexican States, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA publishes draft guidances of two ICH Annexes; EMEA sets format for compliance advice; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration announced a new program regarding the issuance of warning letters to the pharmaceutical industry.

Equipment and Processing Report
The US Food and Drug Administration?s Draft Guidance for Industry?Process Validation: General Principles and Practices provides a life-cycle approach for validating pharmaceutical processes and aims to help pharmaceutical companies achieve consistently high product quality.

Uncovering the root cause of contamination and leveraging the collaborative learning loop of QbD -Aegis Whitepaper