
ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention posted a proposed revised standard for what should and should not appear on the ferrules and cap overseals of medication vials.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention posted a proposed revised standard for what should and should not appear on the ferrules and cap overseals of medication vials.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pharmaceutical Technology talks to FDA's Justina A. Molzon about current discussions with international regulators, and how, after 20 years of harmonization activities and the common technical document, ICH has benefited regulatory authorities.
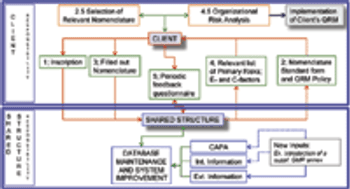
Representatives to the PIC/S provide an example of methodology for implementing ICH Q9.

Dumbed-down presentations and poor speaker selections are destroying a valuable industry tool.

With so many healthcare and pharmacy websites, consumers could use the agency's nod of support.

The authors discuss a study that demonstrates the use of polyethylene oxide mixtures to investigate the effect of polymer viscosity on formulation robustness.

Blame it on cafeteria gossip, outdated procedures, and major miscommunication.

Generic-drug manufacturers look to expand into biologics and complex dosage forms. This article contains bonus online-exclusive material.
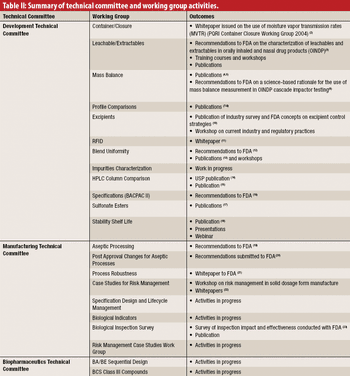
The authors describe the development and organization of the Product Quality Research Institute and highlight some of the important projects conducted by its Working Groups.

Pharmaceutical Technology Europe
The quality assurance environment is forcing pharmaceutical companies to face new challenges. In light of this, the authors conducted a study of professionals from some of the world's top pharmaceutical companies to identify key QA concerns.

Pharmaceutical Technology Europe
Governments, regulators and international agencies continue to defend against counterfeiters; in particular, the European Commission has outlined measures that may help to better regulate pharmaceutical distributors.

Pharmaceutical Technology Europe
Five per cent of consumers across five European countries suspect they may have received a counterfeit prescription and 1% believe that they definitely have, according to research conducted by ICM on behalf of patient safety communications company Aegate (UK).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Society for Chemical Manufacturers and Affiliates (SOCMA) provided an update as to its grassroots efforts regarding the recently passed Chemical and Water Security Act of 2009 (HR 2868).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration has issued 22 Warning Letters to website operators as part of its International Internet Week of Action.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Five percent of consumers across five European countries suspect they may have received a counterfeit prescription, and 1% believe that they definitely have, according to research conducted by ICM on behalf of patient safety communications company Aegate (UK).

ePT--the Electronic Newsletter of Pharmaceutical Technology
Last week, GlaxoSmithKline (GSK, London) agreed to donate 50 million doses of its adjuvanted pandemic influenza A (H1N1) vaccine to the World Health Organization (WHO).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The international pharmaceutical supply-chain consortium Rx-360 held a launch meeting in Europe last week with more than 135 industry and regulatory representatives in attendance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: Genzyme provides updates on enzyme replacement products and FDA complete response letter; Watson appoints VP of global operations; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
On Sunday, Nov. 8, 2009, the Pharmaceutical Research and Manufacturers of America (PhRMA) expressed its disappointment with the US House of Representatives's healthcare-reform bill.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency, the European Center for Disease Prevention and Control, and the Heads of Medicines Agencies issued a European strategy for H1N1 vaccine benefit–risk monitoring.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA and WebMD increase collaboration and PPD is awarded contract to evaluate the agency's postmarket spontaneous-adverse-event surveillance system.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Society of Chemical Manufacturers and Affiliates (SOCMA) reported last week that the US Food and Drug Administration responded to the association's citizen petition relating to the inspection process of foreign drug-manufacturing facilities.

ePT--the Electronic Newsletter of Pharmaceutical Technology
In response to a request from the US Food and Drug Administration, the US Pharmacopeial Convention (USP) revised its standards for propylene glycol and sorbitol solution

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: sanofi aventis forms pact with Micromet; Laureate Pharma adds members to its business team.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Last week, the steering committee and expert working groups of the International Conference on Harmonization (ICH) met in St. Louis, Missouri. A major success of the meeting was the adoption of several annexes to ICH Q4B on pharmacopeial texts.