
To continue innovating, the biopharmaceutical sector needs support from all levels.

To continue innovating, the biopharmaceutical sector needs support from all levels.

As generic divisions become the most-wanted acquisitions of Big Pharma, India's domestic industry may be thinning out.
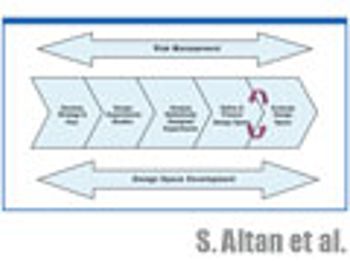
The authors discuss the statistical tools used in experimental planning and strategy and how to evaluate the resulting design space and its graphical representation.

Reports of overlooked controls, dropped pallets, and misplaced documents leave a chill in the air.

After a spate of industrial disasters, the public seeks greater oversight of corporations-so does FDA.

More information may be released to improve public understanding of regulatory policies.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer Suspends Tanezumab Program; Actavis Appoints CEO; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued on June 25 a draft guidance for industry, "CMC Postapproval Manufacturing Changes Reportable in Annual Reports," to provide recommendations to drug applicants about the types of changes that may be included in annual reports.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Office of the Inspector General for the Department of Health and Human Services (HHS) issued a report, Challenges to FDA's Ability to Monitor and Inspect Foreign Clinical Trials, which evaluates the incidence of foreign clinical trials, the ability of the US Food and Drug Administration to oversee these trials, and recommendations to FDA on how to improve its oversight of foreign clinical trials.

Pharmaceutical Technology Europe
Europe needs a more standardised and consistent approach for supplying excipient information to the regulators.

Pharmaceutical Technology Europe
The effects of counterfeiting are hard to measure, both in human impact and financial loss.

Pharmaceutical Technology Europe
The US Pharmacopeia recently convened a meeting of stakeholders to set goals for its 2010–2015 cycle.

Pharmaceutical Technology Europe
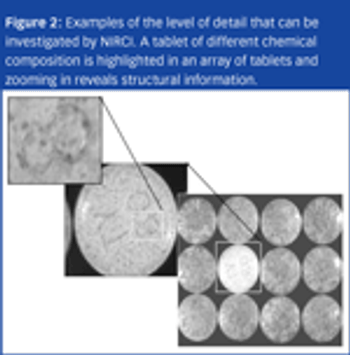
X-ray powder diffraction (XRPD) is a versatile, non-destructive technique that reveals detailed information about pharmaceuticals.

Pharmaceutical Technology Europe
With the increasing financial and technical means of counterfeiters, the number of counterfeit pharmaceuticals in the supply chain is growing at an alarming rate.

Pharmaceutical Technology Europe
The 2D Data Matrix barcode is a familiar sight for most pharmaceutical manufacturers as printing is implemented to meet certain regulations.

Pharmaceutical Technology Europe
We decided to dedicate this month's issue of PTE to a topic that affects all of our readers; the threat of counterfeit medicines.

Pharmaceutical Technology Europe
In order to combat the threat of counterfeits in Europe, the EU's current anti-counterfeiting directive is being amended.
Pharmaceutical Technology Europe
Counterfeit pharmaceuticals are complex products that can vary from their legitimate counterparts both chemically and physically.

Pharmaceutical Technology Europe
Wyatt Earp, the legendary sheriff of Tombstone used to solve troubles in a simple way: aiming, pulling the trigger and bang! ... problem eliminated! Some manufacturers check the readability of their barcodes in the same way.

Pharmaceutical Technology Europe
Teva Pharmaceuticals in the US has admitted to including false statements in the physician prescribing information for its oral contraceptive product Gianvi, a generic version of Bayer Healthcare?s YAZ oral contraceptive.

ePT--the Electronic Newsletter of Pharmaceutical Technology
A study conducted by the European Medicines Agency (EMA) has shown that statistical methods using EMA's EudraVigilance database of adverse drug reaction reports can be used to detect drug safety issues "significantly earlier" compared with routine pharmacovigilance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Regulatory Roundup: USP and Russia's Roszdravnadzor Sign MOU

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration began a partnership with the website Drugs.com to expand access to the agency's consumer-health information.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US House of Representatives held hearings last week to gain testimony on the potential benefits and risks associated with synthetic biology and synthetic genomics.

ePT--the Electronic Newsletter of Pharmaceutical Technology
About one month after the announcement of McNeil Consumer Healthcare's recall of children's liquid pain and allergy medications , the US Food and Drug Administration testified before the US House Committee on Oversight and Government Reform about the issue.