
The advantages of fixed-dose combinations are well recognized but their formulation and manufacture can be a challenge, Stefania Barzanti from IMA Active explains why.
Adeline Siew is editor for Pharmaceutical Technology Europe. She is also science editor for Pharmaceutical Technology.

The advantages of fixed-dose combinations are well recognized but their formulation and manufacture can be a challenge, Stefania Barzanti from IMA Active explains why.

Equipment and Processing Report
The biotech boom, niche markets, smaller batch sizes, and high potency manufacturing are among the key trends shaping the pharmaceutical industry, and equipment must keep up with features such as fast changeovers and automation.

Biotech boom, niche markets, smaller batch sizes and high potency manufacturing are among the key trends shaping the pharmaceutical industry of the 21st century, according to Christian Treitel from Bosch Packaging Technology.

Industry experts share insights on the advances in tablet coating technologies and the potential of continuous coating in solid-dosage manufacturing.
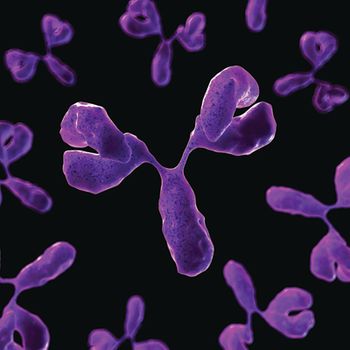
The development of successful ADCs involves careful selection of drug, antibody, and linker, as well as choosing the right attachment chemistry to link the cytotoxic to the antibody.

New formulations that enhance bioavailability, optimize drug-delivery profiles, reduce dosing frequency, or improve patient experience have the potential to deliver quicker returns on investments than developing a completely new drug.

Hedley Rees, managing director at PharmaFlow Ltd and a member of the CPhI expert panel, shares his views on pharmaceutical supply chain issues.

There is growing interest in the development of liquid formulations in prefilled syringes and autoinjectors, which offer convenience and ease of administration in a home setting.
Matt Shaffer, manager, formulation development, Bend Research, a division of Capsugel’s Dosage Form Solutions business, and Martin Koeberle, PhD, head of analytical development and stability testing, Hermes Pharma, spoke to Pharmaceutical Technology about the different approaches used in taste-masking and the challenges involved.

This special issue combines technical articles on leading pharma industry topics and a comprehensive preview of the upcoming CPhI Worldwide trade show

Experts from Dow Pharma & Food Solutions discuss the versatility of solid dispersions in solubility enhancement and the importance of pairing the active pharmaceutical ingredient to the optimal polymer.

Andrew Bulpin, executive vice president of EMD Millipore’s Process Solutions Business, speaks to Pharmaceutical Technology about the trends shaping the biopharmaceutical manufacturing industry.

Randy Wald, senior research fellow at Bend Research, part of Capsugel Dosage Form Solutions shares insights on trends shaping the evolving solid dosage manufacturing landscape, including advances in excipients and the impact on formulation development and manufacturing processes.

Modern methods and modeling offer a better way to understand solubility issues and solve today’s complex formulation challenges.
Prodrugs and drug-delivery systems controlled by time, pH, and osmosis, are being used to prevent drug degradation in the stomach and large intestine and ensure drug release in the colon.

It’s the last day at ACHEMA! With a few more hours left before the show draws to a close, here’s a brief summary of the key trends observed during the week.

Experts at Eppendorf discuss common challenges in cell culture and share insights on possible solutions.

With drug development trends shifting towards personalized medicines, new technologies are needed for the manufacture of these highly sensitive drug products.

Steve Osborn, product design manager at tablet tooling manufacturer I Holland, spoke to Pharmaceutical Technology Europe about the key considerations for successful implementation of multi-tip tooling in tableting operations.
PAT holds the key to real-time quality assurance and consistent product quality in pharmaceutical manufacturing.

Pharmaceutical companies are constantly aiming for shorter drug-development cycles and advances in formulation development produce significant benefits.
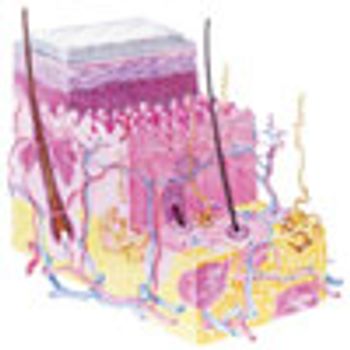
Advances in transdermal drug delivery, particularly with microneedles, are enabling a wider range of drugs to be delivered through the skin.

Endolysin technology targets unwanted bacteria, including resistant strains

WHO says that results from clinical trials should be reported within 12 months of completion of the study

The growing threat and spread of antimicrobial resistance continue to ring alarm bells worldwide.

This is the first time a single strategy document for both EMA and HMA is presented, reflecting the need for a coordinated approach to address the challenges and opportunities facing the European regulatory system network.

CPhI report notes that the Indonesian pharma market is on the brink of a regional manufacturing boom, with market capitalizations and company values rising.
Emerging controlled-release technologies could lead to more effective therapies in the near future.

Pharmaceutical Technology Europe
A review of some of the latest packaging and drug-delivery innovation presented at Pharmapack Europe.

While the skin offers an alternative route of administration for local and systemic drug delivery, developing semi-solid dosage forms can be a challenge.