
Every now and then, you will hear about the rising costs of R&D, which in turn, translate into high prices for new drugs entering the market.
Adeline Siew is editor for Pharmaceutical Technology Europe. She is also science editor for Pharmaceutical Technology.

Every now and then, you will hear about the rising costs of R&D, which in turn, translate into high prices for new drugs entering the market.

The pharmaceutical supply chain continues to be a challenge for the industry despite advances in QbD and PAT.

CPhI experts make predictions on pharma trends for 2015.

PTSM: Pharmaceutical Technology Sourcing and Management
Experts review considerations for selecting excipients for solid-dosage drug performance and manufacturability.

Understanding and preventing protein aggregation is crucial to ensuring product quality and patient safety.

No evidence that Fluad vaccine caused deaths in Italy, says EMA.

Generic drug manufacturing is no longer the only driver for growth in India's pharmaceutical market as more companies start investing in R&D.

Indian manufacturers are moving towards high-value, low-volume work, with complex chemistry and intellectual property challenges.

Gerresheimer, a company with expertise in pharmaceutical and medical technology, announced that it is applying an integrated mold qualification and validation concept for a fast and cost-efficient time-to-market. The company notes that this approach provides process windows for reliable large-scale production and offers options for different deployment scenarios and qualification levels.

Pharmaceutical Technology Europe spoke with Piero Iamartino, R&D director at Micro-Macinazione, about the role of micronization in pharmaceutical manufacturing.

Weighing the pros and cons of hot-melt extrusion and spray drying.

PTSM: Pharmaceutical Technology Sourcing and Management
Industry experts examine reasons why the quality and composition of excipients can vary including environmental factors, processing methods, raw material quality, manufacturing location changes, and operator actions.

What to expect at Paris Nord Villepinte, France as CPhI Worldwide takes off.

An opportunity to broaden your horizon on the latest and hottest industry trends that shape the pharmaceutical landscape.

The countdown has begun for one of the world?s largest pharmaceutical networking events.

The quality and composition of excipients can vary due to environmental factors, processing methods, raw material quality, manufacturing location changes, and even operator actions.

The significant contribution that generic drugs make to health services across Europe cannot be denied.

Increasing concerns are being expressed by industry observers and top-tier pharmaceutical manufacturers that contract manufacturing organizations (CMOs) will not be ready to comply with emerging track-and-trace requirements.

Experts from Capsugel and Catalent discuss the rationale of using lipid0basd formulations to improve the oral bioavailability of poorly soluble drugs.

Pharmaceutical Technology Europe
Pharmaceutical Technology Europe marks its 25th anniversary.

Industry experts share their perspectives on key advances in controlled-release drug delivery and future innovations in this arena.

Industry experts discuss how extractables and leachables studies are designed using a risk-based approach.
Common challenges and key considerations when developing a freeze-drying cycle for protein pharmaceuticals.

Experts share insights on how product appearance such as size, shape, color, and imprint can make a difference in reducing medication errors.

Mark D. Kramer talks about combination products from a regulatory perspective and explains the implications of FDA's final rule on current GMP requirements for combination products.
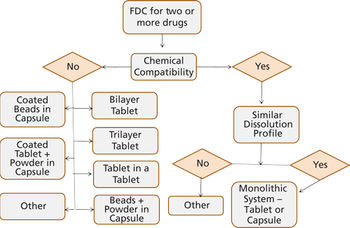
Anil Kane from Patheon spoke to Pharmaceutical Technology about the challenges in the development of FDCs and discussed how to integrate quality-by-design (QbD) in the manufacturing process of FDCs.

Despite the progress made in nanomedicine development, there are several issues that remain unresolved. Raj Bawa, PhD, provides insight.

Nanomedicines can offer promising benefits in the diagnosis and treatment of diseases but there are still concerns about the safety and quality of these therapeutics partly because of the lack of clear regulatory guidance. Scott McNeil, PhD, director of the Nanotechnology Characterization Laboratory (NCL), spoke with Pharmaceutical Technology about nanomedicine characterization.

With nanomedicines on the rise, a new class of non-biological complex drugs (NBCDs), which include nanosimilars, has emerged. As drug regulators are faced with the challenge of defining a framework to ensure the safe introduction of the follow-on nano-therapeutics, Stefan Muhlebach explains why NBCDs cannot be assessed using the standard generic or biosimilar approaches.