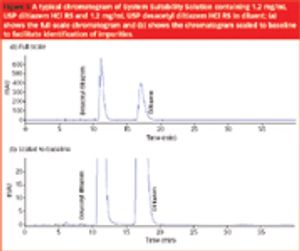
The stability of drugs in solid forms such as powders, tablets and capsules is usually determined very thoroughly by the drug's manufacturer. Based on stability study data, the shelf-life of a drug substance or a drug product is assigned. In addition to dispensing solid dosage forms to patients, pharmacists are frequently asked to compound oral liquid preparations for which the shelf-life or beyond-use date are assigned based on the pharmacist's best judgment - often without the benefit of stability data.