The tightening of intellectual property rights in India under GATT/TRIPS was a crucial inflection point for pharmaceutical outsourcing in India.

The tightening of intellectual property rights in India under GATT/TRIPS was a crucial inflection point for pharmaceutical outsourcing in India.

Risk management is essential in any successful outsourcing partnership. The author outlines the steps toward identifying, understanding, and controlling risk in key manufacturing areas.
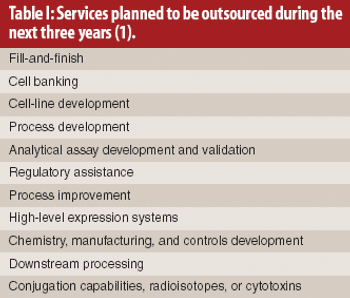
The authors analyze the supply–demand trends for contract biologics manufacturing and the strategies of pharmaceutical and biotechnology companies and their suppliers in the value chain.
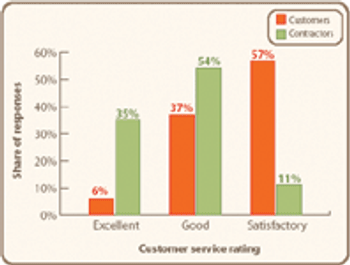
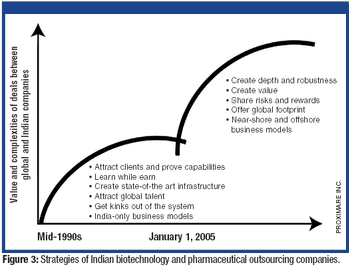
A booming market may be stressing the service-delivery system.

This article is written to assist clinical manufacturing representatives at pharmaceutical companies who are faced for the first time with outsourcing the manufacture of clinical supplies. The author describes the identification, writing, and execution of documents required to support the contract manufacture of products for clinical studies.
Infrastructure must be in place before jumping into outsourcing.
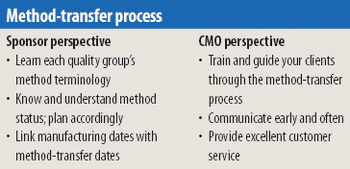
Using basic project management tools and ideas in the transfer process saves time and money and ensures high success rates.
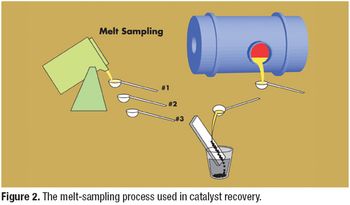
Catalysts are important tools in the synthesis of active pharmaceutical ingredients. Recovery of precious-metal catalysts from a pharmaceutical manufacturing process is a factor in cost control and environmental compliance.