
The reopened debate over embryonic-stem-cell research could stifle many other scientific pursuits.

The reopened debate over embryonic-stem-cell research could stifle many other scientific pursuits.
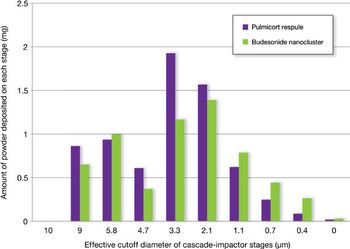
The authors describe a technique designed to yield low-density powders with a tailored particle-size distribution over a broad range of respiratory flow rates.

A recently released industry guide outlines a science- and risk-based approach to control the risk of cross-contamination.
The growth of biologics is an important factor for the injectable-drug delivery systems market. A look at the technical and market considerations affecting this sector.

Private companies and universities are developing new ways to deliver protein drugs.

Public-private R&D partnerships are on the rise across Europe, but national goals and academia-industry competition could prevent their success at the European level.

Global pharmaceutical companies could have a problem getting rid of redundant facilities.

Editors' picks of pharmaceutical science and technology innovations.

An expert-panel-written book has surprising shortcomings.
Drugmakers hatch new manufacturing paradigms in the wake of the 2009 H1N1 influenza pandemic.

The authors provide a brief overview of the European pharmaceutical regulation with regard to batch certification by a qualified person and batch release for the European market.

From disagreement to denial, being cordial about quality control can be challenging.

Pfizer's acquisition of Wyeth involves augmenting its biopharmaceutical product portfolio, pipeline, and related development and manufacturing capabilities.

The president of Gibraltar Laboratories, Daniel Prince, discusses industry trends and challenges.

A look at MVI's malaria work in developing countries.

Novartis' Matthew Stober discusses vaccine manufacturing, including egg- and cell-based systems.

In light of compendial changes, representatives of the US Pharmacopeia and an industry consortium provide perspectives on cap and ferrule labels.
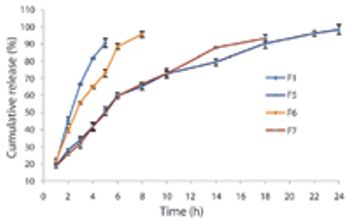
The authors developed a formulation for effervescent gastroretentive drug delivery techniques using ibuprofen as a model drug. They optimized the formulations by applying full factorial design.

President Obama and HHS eye innovation and countermeasures to protect public health.

The EDQM inspection program helps to ensure the quality of APIs on the European market.

Vaccine Manufacturing Reborn