
Ben Venue Laboratories has resumed production on a limited number of manufacturing lines in the company's Bedford, Ohio, facilities.

Ben Venue Laboratories has resumed production on a limited number of manufacturing lines in the company's Bedford, Ohio, facilities.

Watson Pharmaceuticals has received clearance from the Federal Trade Commission for its acquisition of generic drug manufacturer, Actavis.

Dr Reddy's is planning to acquire the specialty injectable company OctoPlus for approximately EUR 27.4 million ($35.7 million) in cash to strengthen its technological capabilities in drug delivery.

Catalent Licenses NJIT Taste-Masking Technology; MicroConstants Completes FDA Inspection; and More.

EMA releases guideline on medicinal products for the treatment of schizophrenia.

Equipment and Processing Report
Consider the time and expertise needed to maintain regulatory compliance and perform stability testing.

Teva has invested $110 million in a new sterile plant in Hungary. The company hopes the plant will strengthen the company's role in Hungarian drug development.

New US Pharmacopeial Convention standards provide a universal approach to organizing labels for prescription containers dispensed by US pharmacists in an effort to improve patient understanding.

At this week's AAPS 2012 Annual Meeting in Chicago, graduate students across the country are being honored for their research and work in bio/pharmaceutical innovation. Pharmaceutical Technology had the chance to talk with a few of the recipients.

GlaxoSmithKline has outlined the measures it is taking as part of a new open innovation approach to R&D, including opening up its tuberculosis compound library, investing in its open laboratory in Spain, and sharing detailed clinical trial data with researchers.

Novasep announced that it will be investing EUR 30 million ($39 million) to build what will be the world's largest chromatography plant for the production of large volumes of commercial APIs.

Mayne Pharma to Acquire Metrics; Sanofi, Bristol-Myers Squibb Restructure Plavix Alliance; and More.

Merck has announced plans to close its headquarters in Whitehouse Station, New Jersey, and relocate to its existing facility in Summit, New Jersey, beginning in 2014.

A national fungal meningitis outbreak linked to contaminated vials of steroid injectables from a Massachusetts compounding operation has reignited the debate over the safety of compounded drugs and the need for stronger FDA regulation of these activities. Last week, the Centers for Disease Control and Prevention reported seven deaths and 91 ill.

FDA announced that it is working closely with the Centers for Disease Control (CDC) to identify the source of an outbreak of meningitis among patients who had received an epidural steroid injection.

Takeda's US subsidiary, Takeda America Holdings, has agreed to acquire the vaccine specialist company LigoCyte Pharmaceuticals for an upfront payment of $60 million in a move intended to bolster and expand the company's vaccine business.

Merck Returns Brinavess Rights to Cardiome; Sigma-Aldrich Expands in Scotland; and More.

Merck Serono's Asceneuron is the third company to be spun off from the company's Entrepreneur Partnership Program that was launched to mitigate the impact from recent restructuring at its Geneva site.

PTSM: Pharmaceutical Technology Sourcing and Management
Several Big Pharma companies strengthen their manufacturing presence in Russia.

New European Pharmacopoeia chapter aims to resolve problems with applying the harmonized UDU test to large sample sizes.

Will the next US President support the backbone of our industry?

Foreign firms struggle against stricter patent laws, but all is not lost.
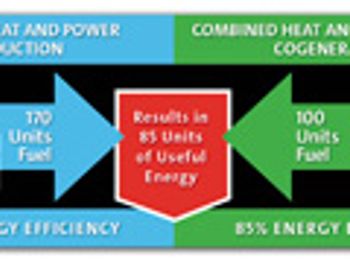
Today's pharmaceutical companies are striving to reduce costs and maximize efficiencies, and must make decisions on the best way to deploy their limited resources.

Only the strong survive when it comes to pharmaceutical packaging and shipping.

Working together affords many unseen opportunities for pharmaceutical innovation.

Recent news stories have reported that FDA scientists have been suspected of leaking confidential, commercial, and trade secret information to the media.

FDA launches BeSafeRx program and releases guidance for industry.

Q&A with David Elder and Richard Wright of Strategic Compliance Consulting, PAREXEL International. Both Elder and Wright formerly served with FDA.

A new report has forecast that the global biopharmaceutical market will be worth more than $320 billion by 2020, up from just $138 billion in 2011.

The European Medicines Agency has updated its guidance on biosimilar medicines, with the aim of helping companies to avoid unnecessary repetition of clinical trials.