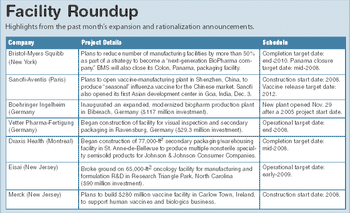
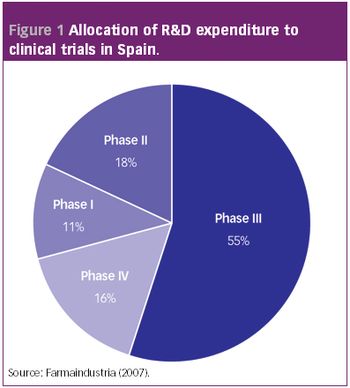
ePT--the Electronic Newsletter of Pharmaceutical Technology
Members of Congress have strongly urged the US Food and Drug Administration to reconsider its proposed rule to amend regulations permitting companies to promptly update their drug and device labels with new safety information.