
The 2011 show presents ideas for package designs and equipment options for packaging lines.

The 2011 show presents ideas for package designs and equipment options for packaging lines.

Indian manufacturers are not a near-term threat to Western CMOs, but may be long term.

A revised book on process analytical technology could be valuable to novices and experts.

Brazil develops its first national plasma fractionation plant.

A single, global tooling standard would offer many benefits, but one has been slow to emerge.

The author describes key considerations for drug manufacturers when evaluating packaging partners.
A new audit guide aims to improve supply-chain security and supplier qualification practice.
The author provides an overview of QbD implmentation for biopharmaceuticals.

Just when things seem to be looking up, the unexpected problem occurs.
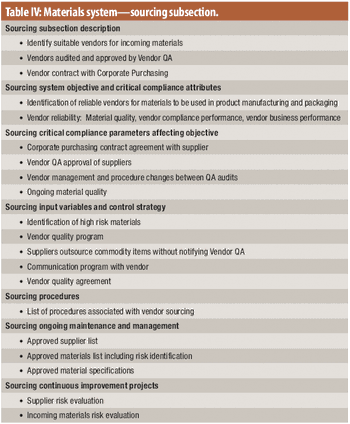
The authors review a compliance-by-design approach to quality systems.

Pharmaceutical Technology's annual survey on equipment and machinery reveals the spending levels and type of spending made in 2010 and planned for 2011.

As drug shortages make headlines, FDA tests the Sentinel safety system and its efect on healthcare.

INTERPHEX 2011 aims to address the industry's unique characteristics.

The Society of Chemical Manufacturers and Affiliates (SOCMA), in a statement submitted to the US House Subcommittee on Cybersecurity, Infrastructure Protection and Security Technologies, called for Congressional action to pass a three-to-five year authorization of current Chemical Facility Anti-Terrorism Standards (CFATS), which are scheduled to expire Mar. 4, 2011.

Gilead to Acquire Calistoga; Bayer Healthcare Appoints Former Pfizer Exec; and More.

HHS Releases a New National Vaccine Plan

President Barack Obama released his budget proposal for fiscal year (FY) 2012, which shows an increase in funding for the US Food and Drug Administration.

EMA Outlines Activities for 2011, and more.

US Senators Amy Klobuchar (D-MN) and Bob Casey (D-PA) introduced the "Preserving Access to Life-Saving Medications Act," which is intended to help address and prevent shortages of prescription drug medications.

Equipment and Processing Report
My drug failed a light-obscuration test, but after I filtered the drug for microscopic particle counting, I couldn?t see any significant particulate on the filter. What might have caused the failure, and why is it not visible?

Equipment and Processing Report
Developments during the past several years have led many industry professionals to question whether nonsterile dosage forms are being produced in a state of control.

Pharmaceutical Technology Europe
The recent India–EU summit was expected to bring news about the upcoming Free Trade Agreement between the EU and India; however, a dispute regarding the supply of generic drugs from India to developing countries has taken centre stage.

Pharmaceutical Technology Europe
India is set to become a "biosimilar powerhouse", with manufacturers in this country well placed to capitalize on the growing biosimilars market, according to market analysts at Datamonitor.

According to the UK's Economic and Social Research Council (ESRC), the closure of Pfizer's UK R&D facility, announced last week, is part of a long-term decline in drug development that is affecting all major UK pharmaceutical multinationals.

On Feb. 4, 2011, the European Medicines Agency released new document on drug products that are manufactured using stem cells.