
Equipment and Processing Report
Normal industrial practice is to control foot-borne contamination with adhesive peel-off disposable mats, but polymeric contamination-control flooring is becoming increasingly popular.

Equipment and Processing Report
Normal industrial practice is to control foot-borne contamination with adhesive peel-off disposable mats, but polymeric contamination-control flooring is becoming increasingly popular.

Eli Lilly Acquires Avid Radiopharmaceuticals; EMA Recruiting New Director; and More

Pfizer (New York) announced on Oct. 29, 2010 that it intends to recall two additional lots--approximately 38,000 bottles--of Lipitor (atorvastatin calcium) 40 mg tablets distributed in the United States.

From fiscal year 2007 to 2009, the US Food and Drug Administration increased the number of foreign drug inspections it conducted, but the agency still conducted fewer foreign inspections than domestic inspections each year, according to a recent report by the US Government Accountability Office (GAO).

FDA Holds Biosimilars Public Hearing

USP is working to ensure quality standards and to increase public information.

IP rights and levels of innovation have opened a bit of controversy regarding decisions being made by Indian courts and legislators.

A conversation with Mike de la Montaigne, president of Eisai Machinery, USA Inc., about the possibilities for conducting fully automated product inspections.

As technology advances, industry's needs are growing.

Scientists and practitioners must work together for the overall good of the patient.

A new center may provide evidence for improving care, but could discourage coverage of treatments.

A Q&A with officers of the departments of State Food and Drug Administration, China, moderated by Ji Xie.

Drug manufacturers have to be more than just "audit ready."

Remaining calm, cool, and collected during mergers and inspections is a feat in itself.
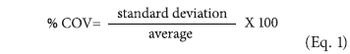
The author examines the process of method development, with reference to ISO 13320:2009 and relevant monographs from the United States and European pharmacopoeias.

The authors present an update to the Wyeth/BASF experience with the IPEC Novel Excipient Safety Evaluation Procedure.

Applied statisticians are forever searching for the enemy of quality-variability.

Pharmaceutical Technology Europe
There has been increasing cooperation between the EMA and the FDA in recent years, and this looks set to continue in the future.

ePT--the Electronic Newsletter of Pharmaceutical Technology
PPD and Bend Research Form Collaboration; Ricerca Makes Senior Appointment; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued a draft guidance last week on the Qualification Process for Drug Development Tools.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeia (USP) has announced two new collaborations.

As part of a team, you will prepare meetings of European Pharmacopoeia groups of experts, contribute to the preparation of working documents for these groups, contribute to the elaboration and revision of European Pharmacopoeia texts and follow up meetings with a view to publication of scientific texts and memoranda.

As part of the Certification team, you will evaluate dossiers submitted with requests for revision of certificates of suitability to the European Pharmacopoeia and prepare draft reports; you will ensure consistency of assessment reports for similar products and consistency of the reports with the policy of the procedure, if necessary analysing and reporting any discrepancies.

ePT--the Electronic Newsletter of Pharmaceutical Technology
During Oct. 5-12, 2010, more than 40 countries cooperated in an international week of action to combat the online sale of counterfeit and illegal drugs and to raise awareness of their associated health risks.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Generic medicines Association (EGA) has made a number of recommendations on how to ensure the quality of medicines in the globalized supply chain; in particular, the EGA spoke about API quality and quality supervision, and how these can be improved by transparent communication between authorities and industry.