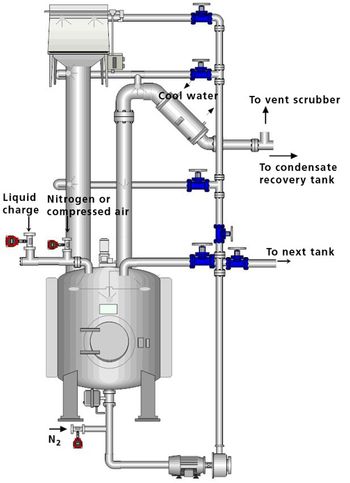
Equipment and Processing Report
The author describes the benefits and challenges inherent to cleaning in place (CIP). The article also describes the development and validation of a CIP cycle.

Equipment and Processing Report
The author describes the benefits and challenges inherent to cleaning in place (CIP). The article also describes the development and validation of a CIP cycle.

AstraZeneca agreed to settle a sex-discrimination lawsuit by paying $250,000 to 124 women who worked at the company's Philadelphia Business Center.

The US Supreme Court ruled in favor of Roche in a patent-dispute case the pharmaceutical company had with Stanford University in a 7-2 vote.

The European Public Health Alliance has called for greater pricing transparency, as well as the formation of a public website that provides comparative information on medicines' procurement prices.

FDA Issues Final Guidance to Amend IND Reporting Requirements.

Ensuring compliance though increased statistical knowledge and resources.

An independent report released by the European Medicines Agency highlighted a number of recommendations to aid the agency in its communication of the benefits and risks of medicines.

Why SOPs are rarely followed, often cited, and in need of follow-through.

Industry struggles to curb drug abuse, diversion, and disruptions in supply.

Sometimes, there are just too many cooks in the kitchen.

FDA added a searchable database of inspection data to its website. The database lists the names and addresses of facilities that the agency inspected during fiscal years 2009 and 2010.
Industry associations will soon provide new recommendations about extractables and leachables.

This month's BIO Convention will encourage needed conversations.

India's drug pricing authority increased the retail costs of certain domestically manufactured drugs, but not those of imported drug products. Pharma is asking why.

Pharmaceutical Technology Europe
The greatest challenge faced by the EMA is the continuing expansion of the EU.

Pharmaceutical Technology Europe
In January 2011, a new version of Annex 11 was released by the European Commission along with a revision of Chapter 4 of its GMP on documentation to reflect the actualities of electronic record keeping, all of which come into full effect in June 2011.

Pharmaceutical Technology Europe
In January 2011, the European Medicines Agency (EMA) announced the new revision of EudraLex Volume 4 (GMP) - Annex 11 'Computerised Systems', and the consequential amendment of EudraLex Volume 4 - Chapter 4 ?Documentation?.

Following recent accusations that the EMA puts companies' commercial interests ahead of public safety, the EMA has responded by emphasizing recent initiatives at the agency to improve data access.

FDA held a joint meeting of its Nonprescription Drugs Advisory Committee and Pediatric Advisory Committee to discuss whether new dosing information for oral over-the-counter drug products containing acetaminophen should be added to the label for children under age 2.

Teva Pharmaceutical Industries agreed to pay shareholders $460 million in cash to acquire a 57% stake in Taiyo Pharmaceutical Industry. Teva also will offer to buy all outstanding shares of Taiyo.

Johnson & Johnson subsidiary Janssen-Cilag International reported that the company is working with regulatory authorities in five countries to address trace amounts of 2,4,6- tribromoanisole identified in five batches of the HIV/AIDS medicine Prezista.

EMA is working with its European and international regulatory partners to monitor and evaluate ?the possible risk of radioactive contamination of medicines manufactured in Japan following the radiation leak from the Fukushima Daiichi nuclear power plant.?

Regulatory approvals for new biopharmaceuticals in the United States have nearly doubled in the past decade compared with the 1990s, says a Tufts study.

Equipment and Processing Report
How do you assign a minimum sample weight for a US Pharmacopeia <41> balance application when the tested repeatability gives a standard deviation of zero?

FDA issued a new guidance for industry concerning the submission of summary bioequivalence data for abbreviated new drug applications.