
The hardest errors to spot are the ones that don't look like errors at all.

The hardest errors to spot are the ones that don't look like errors at all.

Bob Weaver, president of HunterLab, discusses current trends and challenges.

This technical forum is part of a special issue on Solid Dosage and Excipients.
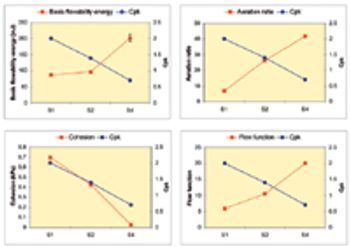
The author explains how to gain an understanding of the relationships between powder characteristics and process performance to match filling-machine geometry to the demands of specific formulations.

The author focuses on how industry can build a system for Total Excipient Control.
Representatives from Pfizer R&D, DEM Solutions, Colorcon, and ARmark Authentication Technologies provide insight into recent tablet-coating technologies.

Pharmaceutical Technology Europe
Europe's approach to pharmacovigilance has room for improvement that European agencies are working on.
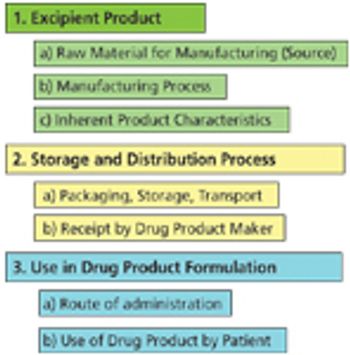
The author describes key considertions for a complete risk-assessment model and provides insight into a pending IPEC guideline in this area.

Nanotechnology often is associated with parenteral drug delivery, particularly for anticancer therapies, but it also has applications in oral drug delivery

Pharmaceutical Technology Europe
The author reviews Warning Letters issued between 2000 and mid-2010 for aseptic processing and non-sterile processing, and determines how many observations were made for each section of the GMPs.

The authors examine an oral-absorption-enhancement technology based on surface-active materials to increase apical membrane fluidity in vitro.

International Conference on Harmonization Hosts a Series of New Training Sessions

On Mar. 23, 2011, sanofi-aventis offered $7 billion in notes to fund its acquisition of Genzyme. The notes are offered in six tranches scheduled to come due between 2012 and 2021, and the interest rates range from 0.05% to 4%.

AstraZeneca has been informed that the UK and US governments? tax authorities have agreed to the terms of an advance-pricing agreement regarding transfer-pricing arrangements for AstraZeneca?s US business for the 13-year period from 2002 to the end of 2014.

US Pharmacopeia Seeks Scientists for its 2011-2012 Fellowship Program

The Bulk Pharmaceutical Task Force (BPTF) of the Society for Chemical Manufacturers and Affiliates (SOCMA) and the European Fine Chemicals Group (EFCG) of the European Chemical Industry Council (CEFIC) are calling on FDA to mandate inspections of foreign active pharmaceutical ingredient (API) manufacturing sites with the cost borne by those sites being inspected. Both organizations have indicated a willingness to pay fees for these inspections when performed on their member-owned facilities that are located outside the United States.

FDA issued a draft guidance for industry on Mar. 11, 2011, titled User Fee Waivers, Reductions, and Refunds for Drug and Biological Products, that offers recommendations to applicants seeking such actions under the Federal Food, Drug, & Cosmetic Act (FD&C Act). A similar draft guidance was issued in July 1993. The new document clarifies the types of waivers, refunds, and reductions available under the user-fee provisions of the FD&C Act as well as the procedures for handling these requests, including how to appeal an FDA decision.

Last Tuesday, the US Senate approved the "America Invents Act," which is intended to reform the nation ’s patent system. If it becomes law, the bill will establish a first-to-file system by defining an invention ’s effective filing date as the actual filing date of the patent or patent application.

FDA and EMA Announce Joint Pilot Program

Last Thursday, FDA filed a consent decree of permanent injunction against McNeil, a subsidiary of Johnson & Johnson, for failing to comply with current good manufacturing practice requirements. The action prevents McNeil from manufacturing and distributing drugs from its Fort Washington, Pennsylvania, facility until FDA determines that its operations comply with the law.

Japan has suspended the use of two pediatric vaccines made by Pfizer and sanofi following the deaths of four children in three days.

AstraZeneca announced this week that it will discontinue the production of Pulmicort (budesonide) 100 and 200 µg/dose HFA (hydrofluoroalkane) pressurized metered-dose inhaler (pMDI) due to manufacturing issues related to technical aspects of the device, which prevents the ongoing manufacture of the product.

FDA Posts PDUFA Meeting Minutes

FDA announced on Mar. 2, 2011, that it is taking action against companies that manufacture, distribute, or market certain unapproved prescription oral cough, cold, and allergy products, according to an agency release.

A number of European public health and transparency campaigners believe that conflict of interest rules may have been breached with the EMA's decision to allow its former Executive Director, Thomas Lönngren, to take up an advisory role within the private pharmaceutical sector.