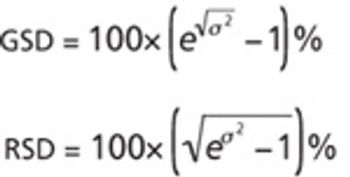
The author argues that traditional concerns about repeatability and intermediate precision remain valid but insufficient.
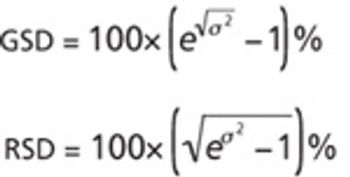
The author argues that traditional concerns about repeatability and intermediate precision remain valid but insufficient.

Those who doubt there's faith in science, should check out our annual Bioprocessing Survey.

FDA, NIH and industry seek new strategies to spur drug development and promote access to therapies.

Monograph modernization and standards donation go hand in hand.

India has the potential to become the new star of the biotechnology industry.

Many factors affect research results.
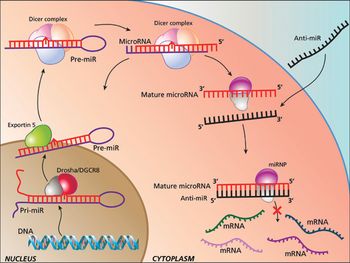
The authors provide further insight into microRNA biology, and the simplicity of anti-miR oligonucleotide drug delivery.

The authors question certain aspects of the industry's current regulatory-compliance strategy and suggest that aseptic-process control and evaluation should be revised.

The author describes recent developments to help overcome the downstream processing bottleneck. This article is part of a special issue on Sterile Manufacturing and Bioprocessing.

Linking peptides to polyethylene glycol, or PEGylation, has helped improve pharmaceutical therapeutics in several ways. A wave of new techniques is now ushering in further advances.

FDA has released a list of its strategic priorities for the next five years to address new global challenges.

The EMA has concluded a class review of bisphosphonates, adopted a number of positives opinions for new medicines and revealed more about the ongoing studies regarding GlaxoSmithKline's Pandemrix pandemic influenza vaccine and a possible link with narcolepsy.

FDA Released Draft Guidance for Industry on Safety Labeling Changes.

Equipment and Processing Report
To find out how well industry is applying QbD, and what benefits the approach can bring, Equipment and Processing Report talked to Moheb M. Nasr, director of FDA?s Office of New Drug Quality Assessment.

FDA recently published guidance for preventing the cross-contamination of finished pharmaceuticals and active pharmaceutical ingredients with nonpenicillin beta-lactam antibiotics.

FDA Released Draft Guidance for Industry on Safety Labeling Changes.

New Web Search Feature on FDA.gov, Search for Product Recalls More Quickly

Johnson & Johnson has instituted a new structure for its Consumer Group according to a Reuters report.


The Society of Chemical Manufacturers and Affiliates criticized the Secure Chemical Facilities Act, which would require chemical facilities to use inherently safer technology as part of chemical-site security measures.

PTSM: Pharmaceutical Technology Sourcing and Management
Indian manufacturers are not a near-term threat to Western CMOs, but may be long term.

Courts and Congress seek to reshape policies and programs.

Research and development may be headed for divorce.

The contract-research industry in China is growing in leaps and bounds, and Big Pharma is leading the way.

Regulators question whether particles that they can't see hurt patients.