Editors' Picks of Pharmaceutical Science & Technology Innovations

Editors' Picks of Pharmaceutical Science & Technology Innovations

CMOs' business model has all of the flaws of the captive model it is meant to replace.

Could President Obama's tax reform, which is targeted at reducing outsourcing, endanger India's contract-services industry?

Emerging markets remain an important element in the strategies of pharmaceutical companies and their suppliers.

Pre-Interphex 2010 Product Releases.
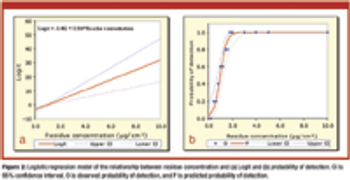
Current methods for establishing visible residue limits (VRLs) are not statistically justifiable. The author proposes a method for estimating VRLs based on logistic regression.
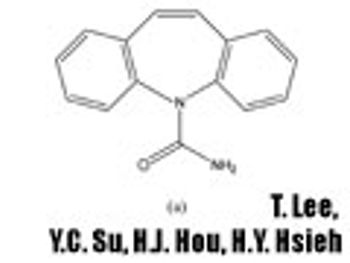
In Part I of this article, the authors describe the materials and methods used in developing a screening strategy to accelerate the preparation and characterization of spherical agglomerates by spherical crystallization.

To best carry out the vision of Hatch-Waxman, Congress must act now on biogenerics.

Contract manufacturers report improving business conditions, but will they continue?

Brief pharmaceutical news items for March 2010.

Analysis of the opportunities and challenges in the biosimilars market.

FDA modernizes information systems, expands access to drug safety and use information.

From last-minute product inserts to putting out fires, close calls are a common occurrence.

It's time to maintain a thorough traceable excipient trail.

If both sides of the aisle don't agree on even mild healthcare reform soon, the bill could die out.

The vice-president addresses shifts in process and more.

Technological developments make it easier to manufacture sterile parenterals.

Visitors to INTERPHEX 2010 will find an abundance of packaging innovations. Plus: Check out our preliminary program for LIVE video interviews at the show.

Pharmaceutical companies and their suppliers share approaches in risk mitigation in sourcing and ways to optimize the outsourced relationship.