
Up Close and Personal

Up Close and Personal

Eastern Europe's pharmaceutical leader, Hungary, is working to maintain its number-one status while also pursuing new avenues, especially in biopharmaceuticals.

Visitors found new container options, child-resistant concepts, and serialization solutions. This article contains bonus online material.

Lessons from the earthquake in Japan show the vulnerability of the bio/pharma supply chain.
Editor's picks of analytical instrumentation products for May 2011.

Regulators and standard-setting bodies are re-examining over-the-counter drugs.
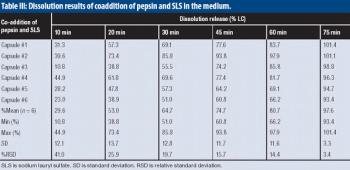
The authors develop a pratical approach to avoid unwanted interactions between pepsin and SLS in dissolution Tier II tests.

Greg Irace, president of sanofi-aventis, on recent industry trends.

The authors examine the influence of glass-transition temperature, melt viscosity, degradation temperature, and process settings.
Efforts are made to educate health workers in less developed countries.

Emerging methods could provide alternative ways of producing inhalable drug particles.

Approaches in using small-molecule and peptide synthesis offer promise in widening the scope of drug candidates.

Nanosponges, a controlled-release nanoparticle system, shows promise in targeted drug delivery

Those who doubt there's faith in science, should check out our annual Bioprocessing Survey.

FDA, NIH and industry seek new strategies to spur drug development and promote access to therapies.

Can the semiconductor industry help Big Pharma develop therapies?

Monograph modernization and standards donation go hand in hand.

Innovator and generic-drug companies need to adapt to compete in the biosimilars market.

India has the potential to become the new star of the biotechnology industry.

Many factors affect research results.

Students and professionals can learn a good deal from a review of single-use technology.