Analytics
Latest News
Latest Videos

Shorts




More News

Counterfeit pharmaceuticals jeopardize public health worldwide, often leading to ineffective or dangerous treatments. Enter Raman spectroscopy: a rapid, non-destructive analytical technique that unveils the molecular secrets of medications. With its ability to identify counterfeit drugs through unique molecular fingerprints—even without unsealing their packaging—this technology is transforming pharmaceutical forensics and safeguarding global health.

Quality, stability, sustainability, and the increased and thoughtful integration of artificial intelligence are foremost in the minds of those on the leading edge of testing trends.

Pharmaceutical Technology® spoke with Dr. Mark Arnold, owner and principal, Bioanalytical Solution Integration, ahead of AAPS PharmSci 360 to find out how bioanalysis enhances bio/pharmaceutical drug development.

Artificial intelligence and machine learning are anticipated to boost success rates.

Artificial intelligence, machine learning, and novel instruments are helping drug developers evaluate complex data from bioanalytical studies.

In the conclusion of this three-part series, the author explores the potential use of agentic AI in pharmaceutical R&D.

In this continuation of a three-part series, the author explores the potential use of agentic AI in pharmaceutical R&D.

Advances in digital technologies offer effective data handling for bio/pharma manufacturing.

In a three-part series, the author explores the potential use of agentic AI in pharmaceutical R&D.

Bothe data integrity and quality of data are critical for drug discovery, manufacturing efficiency, regulatory compliance, and patient safety.

The laboratory services provider is responding to growing demand for packaging and microbiological testing by expanding its laboratory space at its Wiesbaden, Germany, facility.

Survey responses indicate that bio/pharma faces tariff-driven rising costs and supply strain, with firms aiming to boost compliance and diversification and seeking stable trade and R&D support.
The scaled adoption of cell and gene therapies demands a new era of agile, precise, and efficient quality control methods. Manufacturers and diagnostic partners must collaborate to create innovative, compliant testing strategies that preserve product integrity, meet tight timelines, and deliver life-saving treatments faster.

The company presented new data from its Alzheimer’s disease research efforts across its diagnostics and pharmaceutical portfolios at this year’s AAIC.

Help us get a better picture of how new tariffs and trade policy have impacted you and your organization.

The authors performed interference and enhancement testing using a formulated alkaline and acid cleaner, as well as common biopharmaceutical process residues.

EMA is phasing out animal testing and is asking pharmaceutical companies to use new approach methodologies.

Artificial intelligence and machine learning can help identify complex patterns.

ICH Q6B provides expectations and a clear framework for the structural characterization of biopharmaceutical products.
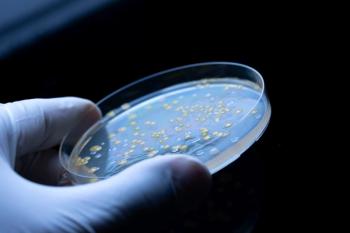
This article explores the impact of test volume, microbial distribution, and dilution errors on bioburden testing variability. It presents statistical approaches to estimate percentage error and discusses strategies to optimize microbial enumeration techniques in biopharmaceutical quality control.

The shift toward personalized medicines poses new challenges in cleanroom protocols.

The authors examine the risks of extractables and leachables, and present solutions that emphasize the importance of a strategic, multi-prong approach.

The CytoFLEX mosaic Spectral Detection Module offers up to 88 channels for detection.

The ‘full tolerance coverage method’ is introduced as a coverage estimation approach for assessing the uniformity of dosage units from large sample sizes, ensuring that no dosage unit exceeds the specification range.

The company now performs product-sterility testing through rapid microbiological methods at two laboratory sites in the United States and one in Germany.