
Takeda has received FDA approval for HYQVIA, a subcutaneous immunoglobulin for the maintenance treatment of chronic inflammatory demyelinating polyneuropathy.

Takeda has received FDA approval for HYQVIA, a subcutaneous immunoglobulin for the maintenance treatment of chronic inflammatory demyelinating polyneuropathy.

Looking at the role that RSV and pediatric medical practices play in drug shortages.

Pharmaceutical Technology Europe® spoke with Joren van der Horst from Luo Automation, a Dutch company focused on automated visual inspection solutions, at the inaugural Start-Up Market on the show floor at CPHI Barcelona 2023.

Pharmaceutical Technology Europe® spoke with Carlos Rial Calvo from FABRX, a pharmaceutical 3D printing company, at the inaugural Start-Up Market on the show floor at CPHI Barcelona 2023.

Pharmaceutical Technology Europe® spoke with Gautam Shetty from Congruence Medical Solutions, a US drug delivery device company, at the inaugural Start-Up Market on the show floor at CPHI Barcelona 2023.

The newly established business under Advent International and Warburg Pincus will be named Simtra BioPharma Solutions.

Dosage forms are being shaped by numerous factors, with patient-centricity continuing to be an important driver of decisions in development.

The European Commission has granted approval for Tyruko (natalizumab), a biosimilar developed by Polpharma Biologics for treating multiple sclerosis.

Tumour-infiltrating lymphocyte (TIL) therapies offer a new route to target cancer.

European vaccine manufacturing is being boosted with high level investments.

An mRNA conversation about the recent past, current hot topics, and probable future.
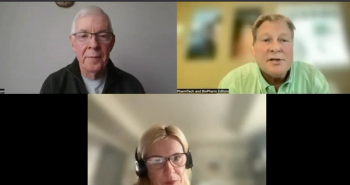
Experts discuss the promise of personalized cancer vaccines through the inherent nimbleness of mRNA platform.
A comprehensive understanding of what makes mRNA vaccines tick is crucial in charting the future of the market.

Current good manufacturing practices for oral solid dosage forms protect the product from contamination and potential errors.

Teva Pharmaceuticals and Alvotech will expand their partnership for the development and commercialization of biosimilar candidates.

Jeff Fischer, co-founder and president of Longhorn Vaccines and Diagnostics, discusses the history and ongoing development of both protein- and mRNA-based vaccines.

In this episode, Sergey Vlasenko from Agilent chats about bio/pharmaceutical manufacturing trends, with a particular focus on oligonucleotides.

When evaluating a drug’s risk assessment for elemental impurities, one must consider all aspects of its lifecycle.
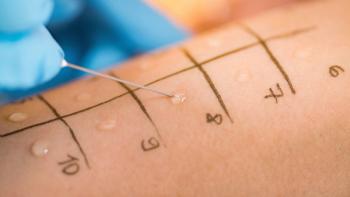
FDA’s new guidance on in-vitro permeation test studies published in October 2022 gives technical and statistical requirements for conducting these tests to compare topical generic drugs with their reference products.

Chris Spivey, editorial director, speaks with Sean Hart, CEO and SCO of LumaCyte, on percent transduction for AAV measured.

invoX Pharma has made a second tranche of investment in pHion Therapeutics to support further development of next-generation mRNA vaccines.

A partnership between Rentschler Biopharma and Ikarovec will support the accelerated development of novel gene therapies for the treatment of eye diseases.

API supplier Olon has started construction of a new manufacturing facility in Milan, Italy, which will be dedicated to production of cytotoxic payloads and linkers for ADCs.

Sandoz’s marketing authorization applications for a proposed biosimilar to denosumab are supported by a comprehensive analytical and clinical data package.

Alvotech and Advanz Pharma have signed a strategic partnership agreement for the supply and commercialization of five biosimilar candidates in Europe.