Dosage Forms
Latest News

Latest Videos

More News
Datwyler has launched new coated plungers in its NeoFlex line that are suitable for large volume biologics.

At AAPS PharmSci 360, Karunakar Sukuru, RPh, PhD, the global vice-president-Rx Product Development, Pharma and Consumer Health, at Catalent Pharma Solutions, discussed lipid-based formulations and tackling bioavailability challenges for oral biologics.

Karunakar Sukuru, RPh, PhD, the global vice-president-Rx Product Development, Pharma and Consumer Health, at Catalent Pharma Solutions, spoke on recent challenges and trends in softgel formulations.
Michelle Logan, vice president of the drug product division for Thermo Fisher Scientific, talked about expanding capability to better serve customers in the OSD market.

This Behind the Headlines news roundup panel discussion on recent news items includes updates regarding sickle cell therapies (e.g,, Pfizer’s withdrawal of Oxbryta and Vertex’s choosing of Lonza as the commercial manufacturer of Casgevy); Emergent BioSolutions $400 million BARDA contract for an mpox vaccine; and the first in a generation novel schizophrenia drug, Cobenfy (xanomeline and trospium chloride). The panel also examines investment revival with ARCH Ventures $3 billion biotech fund raise and the $3.6 billion for Flagship Pioneering. Rounding things out is a discussion on COVID-19-associated childhood myopia and patent thickets artificially keep the price of drugs high.

The announcement of the pairing comes several months after Adare Pharma Solutions said it would upgrade several of its facilities, including in Milan, Italy, where the 3D printing operations will be located.

A coordinated and international response is needed to help control the latest mpox outbreak in Africa.

The ROSS FDA-3500 dual shaft mixer is equipped with independently controlled drives and is designed to efficiently produce good turnover and impart shear to a viscous batch.

The NexGen Press 45 is the latest member of GEA Pharma & Healthcare’s NexGen Press line of tablet presses.

STADA and Alvotech have launched Uzpruvo in Europe, making it the first approved biosimilar to Stelara (ustekinumab) in the European market.

Pelletization, a popular oral drug delivery method in pharmaceuticals and nutraceuticals, offers numerous benefits. This article delves into the critical parameters for formulation development and technological consideration in pelletization, elucidating its significance in advancing pharmaceutical solutions.

The companies have restructured their existing collaboration into a licensing agreement that invests in mRNA development.

Biosimilar analytical assessments focus on demonstrating biosimilarity and interchangeability with the reference biologic.

Pharm Tech Group looks at the challenges surrounding the development and manufacture of ATMPs in more detail with Agne Vaitkeviciene, CEO of Memel Biotech.
A combination of rapid sterility methods with the industry standard compendial method should ensure maximum safety.

A greater understanding of molecular and cellular biology and technological advancements are revolutionizing the field of biologics.

Technological advances are helping advance biologics development and manufacturing and reduce bottlenecks.

Advanced oral dosage forms allow for modulation of controlled- and sustained-release profiles.
Packaging and packaging line innovations are addressing the industry’s growing use of biologics.

In this episode of the Drug Solutions Podcast, Jan Bekker, Vice President of Business Development, Commercial and Technical Operations at BioCina, discusses the latest analytical tools and their applications in the drug development market.

New plans laid out by AstraZeneca will see £650 million ($828 million) be invested into the UK.
Advancements in personalized medicine and other innovations are transforming the way dosage forms are viewed.

This study aimed to develop a taste-masked drug resin complex using the ion exchange resin Kyron T-114.

Given the criticality of fill/finish processes, it is clear that automation is the next technological step.
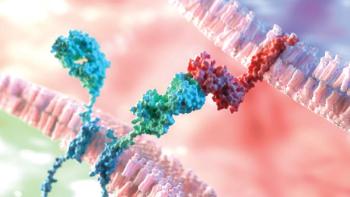
Novel genetic engineering technologies are transforming the design and manufacture of bispecific antibodies, which are emerging as a promising new class of biologics.