
The new partnership between Sandoz, a Novartis division, and Just – Evotec Biologics will aim to develop and manufacture multiple biosimilars.

The new partnership between Sandoz, a Novartis division, and Just – Evotec Biologics will aim to develop and manufacture multiple biosimilars.
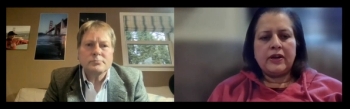
Vibha Jawa, executive director at Bristol Myers Squibb, discusses foreign epitopes recognition from what should be fully human antibody therapies.

The market potential of nucleic acid-based therapies have been pushed into the spotlight following the success of the COVID-19 vaccines.

E&L analytical testing methods can be determined through pointed consideration of the differences small- and large-molecule drug products.

In this episode, Hanns-Christian Mahler and Andrea Allmendinger from ten23 health will discuss some key aspects of biologic drug development and manufacturing.

Unique solutions are required to protect inherently unstable messenger RNA.

Nicole Lewis, PVP Program Manager at Boston Children's Hospital, discusses the science behind precision vaccines, a form of personalized vaccines.

Marcel Baumann, business development director at Recipharm, discusses soft mist inhaler technology from the show floor at the World Vaccine Conference.

Despite the advantages intensified processes can offer developers, there are still some obstacles to overcome before broader application of new technologies will be implemented.

Alternative approaches to freeze-drying are gaining popularity and will help to overcome some of the challenges associated with conventional lyophilization.

Takeda received approval in Japan for a subcutaneous injection formulation of Entyvio (vedolizumab) for treating ulcerative colitis.

Sandoz, a Novartis division, is expected to invest at least $400 million to build a new biologics manufacturing facility in Lendava, Slovenia, to support growing demand for biosimilars.

In this episode, Rhonda Henry and Adam Mendizabal from Emmes discuss the role CROs play in cell and gene therapies undergoing clinical trials at all stages.

This article discusses the evaluation of paired content uniformity and weight variation data sets using F and T tests to ensure that batches meet compendial requirements.

Advancements in therapeutic modalities are necessitating change in drug delivery to help negate some of the expense of development and manufacturing.
EnteroBiotix is harnessing the power of the microbiome to target an array of diseases.

Lilly will cap insulin prices for patients with and without insurance at $35 per month.

Quotient Sciences’ facility expansion is designed to increase support for fully integrated drug development programs.

In this paper, the authors lay out some commonly accepted HCP analytical methods, the challenges of HCP assay, and provide recommendations on what can easily be accomplished in-house and when it may be better to outsource.

Shifting toward more technological solutions and ensuring a greater understanding of the workforce’s needs will give both CROs and sponsors a market advantage.
Efforts to optimize the delivery of sensitive biotherapeutics continue to evolve.

This new pioneering phase of mRNA is loaded with potential, but also obstacles and false dawns.

Materials enhance sustainability and efficiency.

Bio/pharma has evolved and adapted to a variety of challenges in 2022, but what might be on the cards for the industry in 2023?
Manufacturers must consider key components of manufacturing solid versus semi-solid drugs to create a successful end product.