
REMS to improve the safe use of opioids may lead to controls on other high-risk medicines.

REMS to improve the safe use of opioids may lead to controls on other high-risk medicines.

Some GMP agents seem to find a way to squander time, money, and common sense.

Is it good policy to pay for bad behavior?

Traditional Chinese Medicine is widely used, but questions persist regarding its regulatory status.
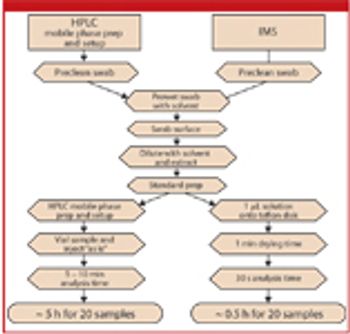
The authors discuss the theory of ion mobility spectrometry, its benefit over HPLC analysis in cleaning verification, and the experimental considerations for method validation and validation.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Physician Payments Sunshine Act is still pending before the US Senate Committee on Finance, according to Jill Kozeny, communications director for Senator Chuck Grassley (R-IA).

ePT--the Electronic Newsletter of Pharmaceutical Technology
Former Senator Majority Leaders Howard Baker, Tom Daschle, and Bob Dole released a report this month, Crossing Our Line: Working Together to Reform the US Health System, which proposes four pillars of health reform.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Following the 2008 launch of the European Fine Chemicals Group (EFCG) Voluntary Guidelines (VGs) to promote supply-chain security, the group has now announced the official launch of an assessment template that will allow fine chemicals customers and suppliers to assess and implement this new set of recommendations.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA debars clinical investigators; Jubilant Organosys forms deal with Endo Pharmaceuticals; AMRI makes changes to its India management team; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Last week, the steering committee and expert working groups of the International Conference on Harmonization met in Yokohama, Japan, to discuss pending guidelines and other relevant issues.

Equipment and Processing Report
To address the increasing popularity of drug-injector systems, the US Food and Drug Administration last week released a Draft Guidance for Industry titled, Technical Considerations for Pen, Jet, and Related Injectors Intended for Use with Drugs and Biological Products.
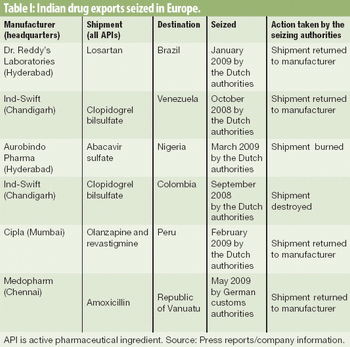
Patent infringement claims and a lack of clear global trade distribution routes may be unraveling the country's generic-drug export industry.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Government Accountability Office recommended that the US Food and Drug Administration draft a plan, including milestones, for developing its Sentinel system and ensuring the privacy and security of patients' healthcare data.

ePT--the Electronic Newsletter of Pharmaceutical Technology
To address the increasing popularity of drug-injector systems, the US Food and Drug Administration last week released a Draft Guidance for Industry titled, Technical Considerations for Pen, Jet, and Related Injectors Intended for Use with Drugs and Biological Products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration released a new guidance for industry, Medication Guides-Adding a Toll-Free Number for Reporting Adverse Events, on June 8, 2009.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The chief operating officer (COO) of Mylan (Canonsburg, PA), one of the world's leading generic drug manufacturers, has condemned the launch of authorized generic drugs during 180-day exclusivity periods.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Commission's (EC) Directorate-General for Enterprise and Industry (EC-DG Enterprise) announced last week that it will not continue preparing a commission directive on good manufacturing practices (GMPs) for certain excipients.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The United Kingdom's Medicines and Healthcare products Regulatory Agency (MHRA) is seeking opinions on a European Commission (EC) proposal that would allow drugmakers to disseminate certain information on prescription medicines directly to European Union patients.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Members of the House Committee on Energy and Commerce, Chair Emeritus John D. Dingell (D-MI), Chair Henry A. Waxman (D-CA), and Reps. Frank Pallone (D-NJ), Bart Stupak (D-MI), Diana DeGette (D-CO) and Betty Sutton (D-OH) released last week a discussion draft of the Food Safety Enhancement Act of 2009.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The International Serious Adverse Event Consortium (SAEC), a nonprofit partnership, has helped the US Food and Drug Administration gain significant understanding of the genetic basis for drug-induced liver injury, according a new FDA press release.

ePT--the Electronic Newsletter of Pharmaceutical Technology
This week the US Food and Drug Administration released the final version of Guidance for Industry: Providing Regulatory Submissions in Electronic Format-Drug Establishment Registration and Drug Listing.

ePT--the Electronic Newsletter of Pharmaceutical Technology
In two whistleblower suits filed in May 2009, the United States and 16 states alleged that Wyeth (Madison, NJ) knowingly failed to offer the government the same discounts it gave to private purchasers of its drugs, as Medicaid laws require.

A book guides readers through the regulatory requirements for computerized quality systems.

A successful quarantine program requires collaboration among all departments of a pharmaceutical facility.

As regulators work to curb counterfeiting, industry finds benefits to gaining granular data about the supply chain. This article contains bonus online-exclusive material.