
FDA recently sent sanofi-aventis two Warning Letters for its facilities in Frankfurt am Main, Germany, and Bridgewater, NJ.

FDA recently sent sanofi-aventis two Warning Letters for its facilities in Frankfurt am Main, Germany, and Bridgewater, NJ.

What to do when your CMO changes the manufacturing equipment line.

Brazil develops its first national plasma fractionation plant.

A single, global tooling standard would offer many benefits, but one has been slow to emerge.

Social media tools have taken over many aspects of our lives, now including regulatory info.
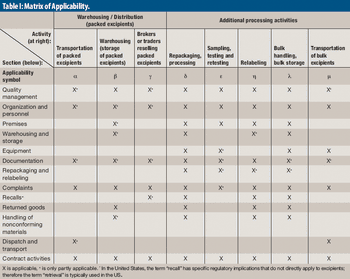
A new audit guide aims to improve supply-chain security and supplier qualification practice.

Just when things seem to be looking up, the unexpected problem occurs.

FDA's efforts to improve access to treatments for rare diseases.

INTERPHEX 2011 aims to address the industry's unique characteristics.

PTSM: Pharmaceutical Technology Sourcing and Management
A roundup of developments in corporate social responsibility and sustainability from the bio/pharmaceutical industry, its suppliers, and other public and private organizations.

PTSM: Pharmaceutical Technology Sourcing and Management
Bristol-Myers Squibb opens a new pediatric HIV/AIDS clinic in Tanzania.

PTSM: Pharmaceutical Technology Sourcing and Management
This article is Part I of a three-part series on biopharmaceutical issues in public health, government, and developing-world markets. Part 1 focuses on drug-development. Part II, which examines manufacturing, appeared in the April 2011 of Sourcing and Management. Part III, which analyzes distribution and administration,appeared in the May 2011 issue.

Pharmaceutical Technology Europe
After reviewing further data on the suspected link between cases of narcolepsy and GlaxoSmithKline's pandemic influenza vaccine, Pandemrix, the EMA's Committee for Medicinal Products for Human Use (CHMP) is still unable to establish a causal relationship.

Pharmaceutical Technology Europe
An initiative based in the Netherlands aims to bring together universities, pharma companies, international research institutions and product development partnerships to help develop new diagnostic tests and medicines for neglected tropical diseases.

Pharma's manufacturing and supply chain needs a "radical overhaul" because it is underused, inefficient and ill-equipped to cope with new types of products that will be coming to market in the near future, according to a report from PricewaterhouseCoopers (PwC).

The Society of Chemical Manufacturers and Affiliates (SOCMA), in a statement submitted to the US House Subcommittee on Cybersecurity, Infrastructure Protection and Security Technologies, called for Congressional action to pass a three-to-five year authorization of current Chemical Facility Anti-Terrorism Standards (CFATS), which are scheduled to expire Mar. 4, 2011.

Gilead to Acquire Calistoga; Bayer Healthcare Appoints Former Pfizer Exec; and More.

HHS Releases a New National Vaccine Plan

Last Wednesday, sanofi aventis agreed to acquire Genzyme for $74 per share in cash, or approximately $20.1 billion.

President Barack Obama released his budget proposal for fiscal year (FY) 2012, which shows an increase in funding for the US Food and Drug Administration.

EMA Outlines Activities for 2011, and more.

Company and People Notes: sanofi to Acquire Genzyme; Lilly Makes Senior Appointments; and More.

US Senators Amy Klobuchar (D-MN) and Bob Casey (D-PA) introduced the "Preserving Access to Life-Saving Medications Act," which is intended to help address and prevent shortages of prescription drug medications.

Equipment and Processing Report
A major pharmaceutical micronizing company consulted Telstar when it needed a mobile, high-containment isolation system suitable for housing 4-, 8-, 12-, or 15-in. micronizing jet-mill systems during the loading, milling, and offloading processes.

Equipment and Processing Report
My drug failed a light-obscuration test, but after I filtered the drug for microscopic particle counting, I couldn?t see any significant particulate on the filter. What might have caused the failure, and why is it not visible?

Equipment and Processing Report
Developments during the past several years have led many industry professionals to question whether nonsterile dosage forms are being produced in a state of control.

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment and Processing Report, reviews the Editor's Picks for the February 2011 edition from Alfa Laval and GE Healthcare.

Pharmaceutical Technology Europe
The recent India–EU summit was expected to bring news about the upcoming Free Trade Agreement between the EU and India; however, a dispute regarding the supply of generic drugs from India to developing countries has taken centre stage.

Pharmaceutical Technology Europe
According to the UK's Economic and Social Research Council (ESRC), the closure of Pfizer's UK R&D facility, announced last week, is part of a long-term decline in drug development that is affecting all major UK pharmaceutical multinationals.

Pharmaceutical Technology Europe
India is set to become a "biosimilar powerhouse", with manufacturers in this country well placed to capitalize on the growing biosimilars market, according to market analysts at Datamonitor.